By monitoring the body’s molecular response to a viral attack, the new method developed by Flatiron Institute researchers and their colleagues can diagnose even asymptomatic patients with 98.4 percent accuracy.
By inspecting the body’s immune response at a molecular level, a research team has developed a new way to test patients for COVID-19. Their method can potentially catch infections a matter of hours after exposure — far earlier than current COVID-19 tests can detect the virus — with near-perfect accuracy. The team describes their innovation, which is still in the early stages of development, in the February 27 issue of the journal Cell Reports Methods.
Most existing COVID-19 tests “rely on the same principle, which is that you have accumulated a detectable amount of viral material, for example, in your nose,” says study lead author Frank Zhang, who worked on the project as a Flatiron research fellow at the Flatiron Institute’s Center for Computational Biology (CCB) in New York City. “That poses a challenge when it’s early in the infection time window and you haven’t accumulated a lot of viral material, or you’re asymptomatic.”
Leveraging mRNA Molecules for Rapid Diagnosis
The new technique is instead based on how our bodies mount an immune response when invaded by SARS-CoV-2, the virus that causes COVID-19. When the assault starts, specific genes turn on. Segments of those genes produce mRNA molecules that guide the building of proteins. The particular blend of those mRNA molecules changes the types of proteins produced, including proteins involved in virus-fighting functions. The new method can confidently identify when the body is mounting an immune response to the COVID-19 virus by measuring the relative abundance of the various mRNA molecules. The new study is the first to use such an approach to diagnose an infectious disease.
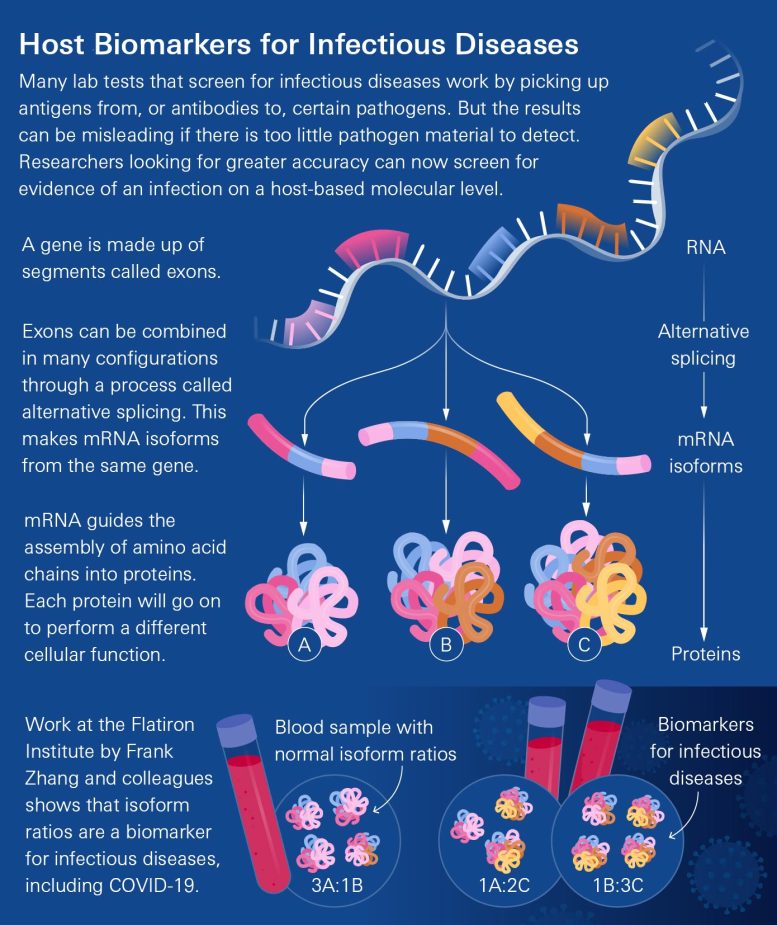
The researchers tuned their method using blood samples from a 2020 study of U.S. Marine recruits taken before and after the participants caught COVID-19. The researchers’ computational framework identified more than 1,000 disease-associated mRNA-variant ratio changes.
Impressive Accuracy in Asymptomatic Cases
When put to the test using real-world blood samples, the new method yielded an impressive 98.4 percent accuracy rating. That’s especially impressive as the approach works just as well on asymptomatic patients, for whom rapid antigen tests can be less than 60 percent accurate. “It was really surprising that it worked so well,” says Zhang, now an assistant professor at Cedars-Sinai Medical Center in Los Angeles. “It’s a promising alternative and complementary approach to conventional PCR tests.”
The new approach isn’t ready for prime time yet, Zhang says. He and his colleagues only tested blood samples rather than the nasal samples that are more common and convenient for diagnosing COVID-19. Also, they need to make sure they can distinguish between the body’s reaction to COVID-19 and its response to infections caused by other viruses, such as colds.
The researchers say they’re optimistic, though, as other research groups have already made progress on tests that look solely at which genes turn on. Those same tests could easily add the mRNA analysis developed in the new study, thereby producing even better results, Zhang says. “Anything they can do, we can probably explore and join forces on,” including catching cases within hours of initial exposure.
Reference: “Blood RNA alternative splicing events as diagnostic biomarkers for infectious disease” by Zijun Zhang, Natalie Sauerwald, Antonio Cappuccio, Irene Ramos, Venugopalan D. Nair, German Nudelman, Elena Zaslavsky, Yongchao Ge, Angelo Gaitas, Hui Ren, Joel Brockman, Jennifer Geis, Naveen Ramalingam, David King, Micah T. McClain, Christopher W. Woods, Ricardo Henao, Thomas W. Burke, Ephraim L. Tsalik, Carl W. Goforth, Rhonda A. Lizewski, Stephen E. Lizewski, Dawn L. Weir, Andrew G. Letizia, Stuart C. Sealfon and Olga G. Troyanskaya, 12 January 2023, Cell Reports Methods.
DOI: 10.1016/j.crmeth.2023.100395
Zhang worked on the computational aspects of the project along with CCB research fellow Natalie Sauerwald and CCB deputy director for genomics Olga Troyanskaya. Stuart Sealfon of the Icahn School of Medicine at Mount Sinai in New York City led the work on the study’s biological components, with Standard BioTools in San Francisco developing the testing setup.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.