Studies using human cell lines and tumors grown in mice provide early evidence that inhibiting RNA-binding proteins, a previously overlooked family of molecules, might provide a new approach for treating some cancers.
In cancer research, it’s a common goal to find something about cancer cells — some sort of molecule — that drives their ability to survive, and determine if that molecule could be inhibited with a drug, halting tumor growth. Even better: The molecule isn’t present in healthy cells, so they remain untouched by the new therapy.
Plenty of progress has been made in this approach, known as molecular targeted cancer therapy. Some current cancer therapeutics inhibit enzymes that become overactive, allowing cells to proliferate, spread, and survive beyond their norm. The challenge is that many known cancer-driving molecules are “undruggable,” meaning their type, shape or location prohibit drugs from binding to them.
A New Frontier: RNA-Binding Proteins
University of California San Diego School of Medicine researchers are now exploring the therapeutic potential of RNA-binding proteins, a relatively untapped family of cancer-driving molecules. After genes (DNA) are transcribed into RNA, these proteins provide an extra layer of cellular control, determining which RNA copies get translated into other proteins and which don’t. Like many molecular systems that govern cell growth, RNA-binding proteins can contribute to tumor development when they malfunction.
In their latest study, publishing today (July 2, 2021) in Molecular Cell, the UC San Diego School of Medicine team discovered in human cells and mouse models that RNA-binding proteins represent a new class of drug targets for cancers, including triple-negative breast cancer, a particularly difficult-to-treat cancer because it lacks most other molecular drug targets.
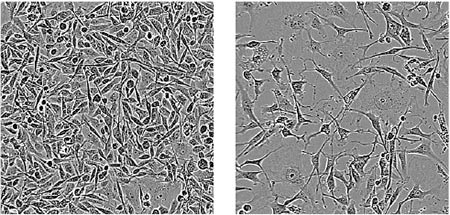
Targeting YTHDF2 Shrinks Tumors
One RNA-binding protein in particular stood out: YTHDF2. When the researchers genetically removed YTHDF2 from human triple-negative breast tumors transplanted into mice, the tumors shrank approximately 10-fold in volume.
“We’re excited that RNA-binding proteins look like they could be a new class of drug targets for cancer,” said senior author Gene Yeo, PhD, professor of cellular and molecular medicine at UC San Diego School of Medicine. “We’re not yet sure how easily druggable they are in this context, but we’ve built a solid framework to begin exploring them.”
Yeo led the study with Jaclyn Einstein, PhD, a graduate student in his lab. Einstein will join a startup company spun out from the lab to explore YTHDF2’s druggability.
Yeo’s team has long studied the role of RNA-binding proteins in a number of other diseases. In 2016, for example, they discovered that mutations in one such protein contribute to ALS by scrambling crucial cellular messaging systems.
A Two-Step Attack on Cancer
To begin exploring RNA-binding proteins as cancer drug targets, the researchers turned to an old philosophy known as synthetic lethality. In this one-two-punch approach, they started with human breast cells engineered to over-produce another well-known cancer-driving molecule, and looked for additional vulnerabilities specific to those cells.
The researchers systematically silenced RNA-binding proteins in these cancer cells one by one using the CRISPR gene editing technique. They found 57 RNA-binding proteins that, when inhibited, kill cancer cells with the known hyperactive cancer-driver. The advantage of the synthetic lethal approach is that normal cells, which don’t produce that cancer-driving molecule, should be left untouched by the treatment. Of these 57 RNA-binding proteins, YTHDF2 appeared most promising.
Yeo’s team also recently developed a new laboratory technique called Surveying Targets by APOBEC-Mediated Profiling (STAMP), which allows them to measure what had previously been largely invisible: how RNA-binding proteins interact with RNA molecules within individual cells.
Triggering Cancer Cell Suicide
The researchers used STAMP in this study to get a detailed look at how the various cells that make up a breast tumor behave without YTHDF2. The approach revealed that YTHDF2-deficient cancer cells die by stress-induced apoptosis, a carefully controlled mechanism cells use to destroy themselves. Apoptosis is supposed to shut down malfunctioning cells so tumors don’t arise, but it doesn’t always work. By removing YTHDF2, they managed to re-activate this cell death signal.
To test how safe it might be to treat cancer by inhibiting YTHDF2, the researchers engineered mice that lack YTHDF2 in every cell of the adult body, not just transplanted breast cancer cells. The mice appeared completely normal — not only did they not have tumors, there were no changes in body weight or behavior.
“Those otherwise healthy mice tell us that we might expect minimal adverse side effects of potential therapies that work by targeting YTHDF2,” Einstein said.
Reference: “Inhibition of YTHDF2 triggers proteotoxic cell death in MYC-driven breast cancer” by Jaclyn M. Einstein, Mark Perelis, Isaac A. Chaim, Jitendra K. Meena, Julia K. Nussbacher, Alexandra T. Tankka, Brian A. Yee, Heyuan Li, Assael A. Madrigal, Nicholas J. Neill, Archana Shankar, Siddhartha Tyagi, Thomas F. Westbrook and Gene W. Yeo, 2 July 2021, Molecular Cell.
DOI: 10.1016/j.molcel.2021.06.014
Co-authors of the study also include: Mark Perelis, Isaac A. Chaim, Julia K. Nussbacher, Alexandra T. Tankka, Brian A. Yee, Assael A. Madrigal, Archana Shankar, all at UC San Diego; Jitendra K. Meena, Heyuan Li, Nicholas J. Neill, Siddhartha Tyagi, Thomas F. Westbrook, all at Baylor College of Medicine.
Disclosure: Gene Yeo and Jaclyn Einstein are inventors on a patent disclosure at UC San Diego related to this work. Yeo is co-founder, member of the Board of Directors, equity holder, on the Scientific Advisory Board and paid consultant for Locanabio and Eclipse BioInnovations. Yeo is a visiting faculty at the National University of Singapore. The terms of these arrangements have been reviewed and approved by the University of California San Diego in accordance with its conflict-of-interest policies.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.