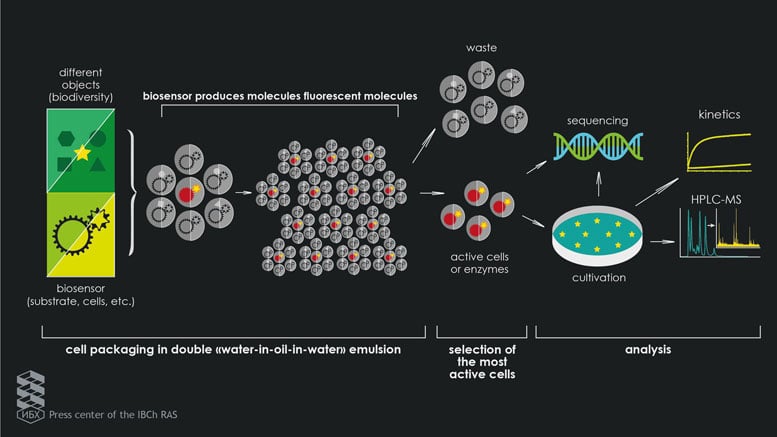
A team of researchers has created a microfluidic system for ultra-high-performance screening in double emulsion droplets. This breakthrough development can be used when studying the unique properties of singular living cells, and is 30 thousand times more productive than robotic workstations. At the same time, it greatly simplifies the work of researchers involved in determining the functionality of biological objects for the creation of medicinal products on their basis.
“Researchers working at various research institutes tend to spend a lot of their working hours trying and testing, purifying and isolating highly-active proteins. Our lab is no exception, and so, we have tried to solve this problem by developing an ultra-high-performance screening system for isolating biomolecules based on microfluidic emulsions. The result is a system that allows us to isolate interesting biological functions from the enormous diversity of any microscopic biological object, not only enzymes,” explains Stanislav Terekhov, Junior Researcher at the Laboratory of Biocatalysis of the Shemyakin&Ovchinnikov Institute of Bioorganic Chemistry of the Russian Academy of Sciences, one of the authors of the article.
The idea came about three years ago when Stanislav proposed developing a technology that allows quickly determining the activity of hundreds of millions of new enzymes produced by his colleague, Ivan Smirnov. Ivan’s working group was engaged in creating and selecting biocatalysts from combinatorial libraries of enzymes that speed up reactions, for which there are no natural enzymes. For example, for the inactivation of organophosphorus toxins, neuromuscular paralytic gases are relevant in connection with the massive spread of the use of pesticides and chemical warfare agents. The researchers had to spend years trying to obtain only a few dozens of new proteins.
Using the photolithography method, which is widely used in technology companies to create computer chips, researchers from the Institute of Bioorganic Chemistry collaborated with colleagues from the St. Petersburg Academic University and the Institute of General Pathology and Pathophysiology and created microfluidic chips with channels that have a thickness smaller than the diameter of a hair to generate the emulsion droplets. The researchers used droplets of “water-oil-water” double emulsion to isolate single cells, allowing them to study their unique properties. Using microfluidic chips, Stanislav and his colleagues put the individual living cells into the droplets, after which the enzymatic and biological activity of the cells in the drops was studied at the Lomonosov Moscow State University using a fluorescence-activated cell sorter. The fluorescence drops helped detect the most active cells, which were used for their selection. The isolated cells in the drops were then further analyzed using classical molecular-biological methods and modern metabolic methods of analysis, as well as large-scale sequencing based on the Federal Clinical Research Center’s physical and chemical medicine.
“As a result, we were able to obtain approximately 108 drops per hour, and in a day, we managed to isolate the necessary amount of enzymes with the required amount of activity,” continues Stanislav. “For example, we managed to improve the enzyme butyrylcholinesterase, which not only linked the organophosphorus toxin, but helped it to hydrolyze, i.e. to destroy it, and link to the next toxin. Afterward, we encapsulated bacterial cells in order to trace which microorganisms are inhibitors of the growth of the highly pathogenic bacteria Staphylococcus Aureus. Thus, our screening method is suitable for discovering new drugs that are enzyme-based or based on microorganisms, their metabolites, and other biological objects.”
This universal microfluidic screening platform that the researchers have developed requires significantly less time and less financial resources. It operates 30 thousand times faster than robotic systems that utilize the services of pharmaceutical companies to search for new drugs, analyzing large libraries of any biological object, including proteins, enzymes and even living cells, in order to search for new and more effective drugs.
Reference: “Microfluidic droplet platform for ultrahigh-throughput single-cell screening of biodiversity” by Stanislav S. Terekhov, Ivan V. Smirnov, Anastasiya V. Stepanova, Tatyana V. Bobik, Yuliana A. Mokrushina, Natalia A. Ponomarenko, Alexey A. Belogurov Jr., Maria P. Rubtsova, Olga V. Kartseva, Marina O. Gomzikova, Alexey A. Moskovtsev, Anton S. Bukatin, Michael V. Dubina, Elena S. Kostryukova, Vladislav V. Babenko, Maria T. Vakhitova, Alexander I. Manolov, Maja V. Malakhova, Maria A. Kornienko, Alexander V. Tyakht, Anna A. Vanyushkina, Elena N. Ilina, Patrick Masson, Alexander G. Gabibov and Sidney Altman, 7 January 2017, PNAS.
DOI: 10.1073/pnas.1621226114
The Kazan Federal University, Skolkovo Institute of Science and Technology, Moscow Institute of Physics and Technology, French Academy of Pharmacy, and Yale University also participated in this groundbreaking research work.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.