
New research shows that brain tissue stiffness helps regulate chemical signals during development. The discovery highlights an unexpected connection between physical forces and the brain’s wiring process.
For decades, scientists have understood that chemical signals, including gradients of signaling molecules, play a central role in directing how tissues grow and organize. More recently, research has revealed that physical factors, such as how stiff or soft a tissue is, also have a direct impact on cell behavior.
What has remained unclear is how these mechanical influences interact with chemical signals to guide development in a coordinated way.
Piezo1 Links Tissue Stiffness to Chemical Cues
Researchers of the Max-Planck-Zentrum für Physik und Medizin (MPZPM), the Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), and the University of Cambridge have uncovered fundamental mechanisms at play in the developing brain. By using Xenopus laevis (African clawed frogs), a well-established model system, the team found that tissue stiffness regulates the expression of key chemical cues and that this process is controlled by the mechanosensitive protein Piezo1.
The team of researchers led by Prof. Kristian Franze found that increasing tissue stiffness induces the expression of chemical signals that are typically absent in those regions. Semaphorin 3A is one such chemical signal. Crucially, this response only occurred when levels of Piezo1 were sufficiently high.
“We didn’t expect Piezo1 to act as both a force sensor and a sculptor of the chemical landscape in the brain,” said study co-lead Eva Pillai, a postdoctoral researcher at the European Molecular Biology Laboratory (EMBL). “It not only detects mechanical forces—it helps shape the chemical signals that guide how neurons grow. This kind of connection between the brain’s physical and chemical worlds gives us a whole new way of thinking about how it develops.”
Reducing Piezo1 levels affected more than chemical signaling alone. The researchers also observed changes in the physical stability of the tissue. Lower amounts of Piezo1 led to reduced levels of important cell-adhesion proteins, including NCAM1 and N-cadherin. These proteins are crucial for maintaining strong connections between neighboring cells, which help hold tissue together.
“What’s exciting is that Piezo1 doesn’t just help neurons sense their environment—it helps build it,” said Sudipta Mukherjee, study co-lead and postdoctoral researcher at FAU and MPZPM. He and Pillai were both doctoral students at the University of Cambridge, where the project was initiated. “By regulating the levels of these adhesion proteins, Piezo1 keeps cells well connected, which is essential for a stable tissue architecture. The stability of the environment, in turn, influences the chemical environment.”
Piezo1 as Sensor and Architect of Tissue
The study suggests that Piezo1 serves two closely linked roles. It acts as a sensor that converts mechanical forces from the surrounding tissue into cellular responses, and it also helps shape the mechanical properties of that tissue by supporting strong cell-to-cell connections.
The implications of this work extend beyond basic brain development. Errors in neuronal guidance are associated with congenital and neurodevelopmental disorders, while abnormal tissue stiffness is known to contribute to diseases such as cancer. By demonstrating that mechanical conditions can actively regulate chemical signaling, the study points to new directions for understanding development and disease.
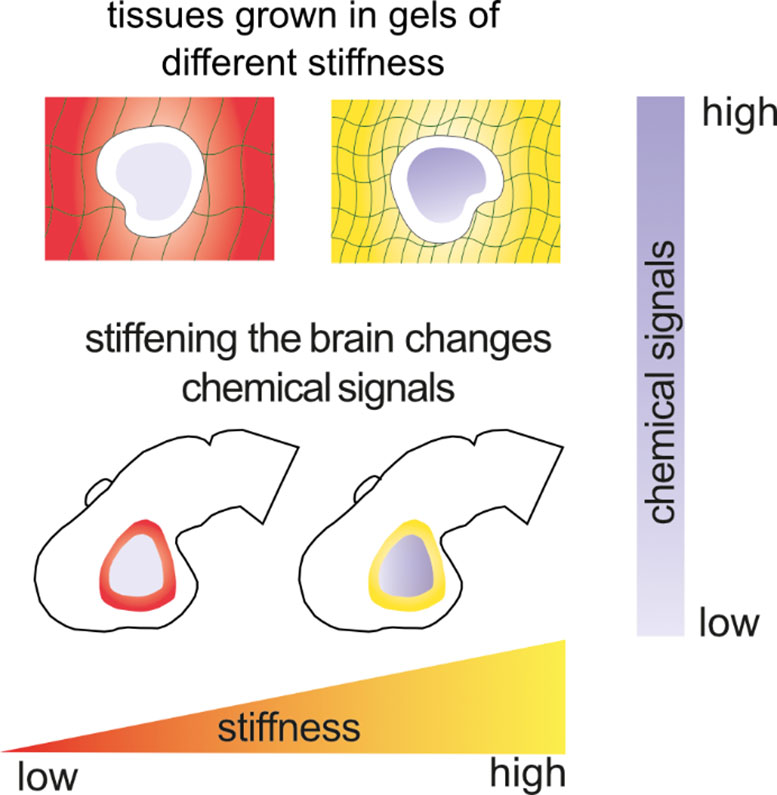
“Our work shows that the brain’s mechanical environment is not just a backdrop—it is an active director of development,” said senior author Kristian Franze. “It regulates cell function not only directly, but also indirectly by modulating the chemical landscape. This study may lead to a paradigm shift in how we think about chemical signals, with implications for many processes from early embryonic development to regeneration and disease.”
The results suggest that changes in tissue stiffness can influence chemical signaling across long distances, affecting cells far from where the mechanical stimulus originates. Together, these findings highlight mechanical forces as powerful drivers of development and organ function.
Reference: “Long-range chemical signalling in vivo is regulated by mechanical signals” by Eva K. Pillai, Sudipta Mukherjee, Niklas Gampl, Ross J. McGinn, Katrin A. Mooslehner, Julia M. Becker, Alexander K. Winkel, Amelia J. Thompson and Kristian Franze, 19 January 2026, Nature Materials.
DOI: 10.1038/s41563-025-02463-9
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.