A receptor involved in normal physiological processes may also drive breast cancer growth when overexpressed.
A surgeon can remove a breast tumor, but surgery does not always eliminate every cancer cell. This is particularly true when the disease has already spread beyond its original location in the body. Scientists are still uncovering the molecular processes that allow breast cancer cells to multiply and travel to new tissues. Researchers at Hiroshima University have now identified one of these mechanisms. Their work also suggests that small chains of protein building blocks could potentially be developed into a new type of anticancer treatment.
The team reported their findings in the British Journal of Pharmacology. Their research centered on vasoactive intestinal peptide receptor-2 (VIPR2), a receptor that binds to two different hormones. Under normal conditions, VIPR2 plays a role in regulating circadian rhythm, immune responses, insulin release, and several other biological processes.
Problems arise when the receptor is overexpressed, meaning that cells produce an excessive amount of it. When this occurs, VIPR2 can contribute to faster breast cancer cell growth and help the disease spread to other parts of the body. When present at high levels, the receptor can also attach to another copy of itself, creating a structure known as a dimer.
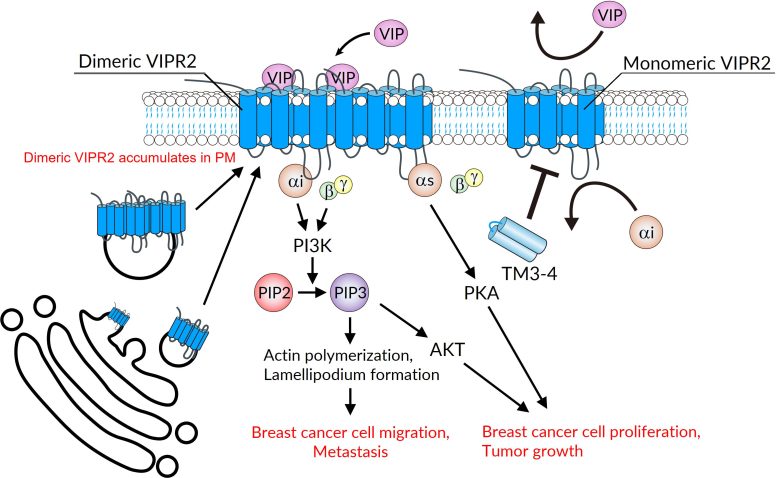
According to co-corresponding author Satoshi Asano, assistant professor in the Department of Cellular and Molecular Pharmacology at Hiroshima University’s Graduate School of Biomedical and Health Sciences, many protein receptors can form pairs with identical molecules in a process called homodimerization. When two receptors combine in this way, the resulting dimer can behave differently from a single receptor unit, which is known as a monomer.
“However, it was unclear whether VIPR2 monomers directly interact with each other and what function the dimeric receptor has in breast cancer cells,” Asano said.
Investigating Receptor Dimerization
In the new study, the researchers first confirmed that VIPR2 molecules can directly connect with each other to form homodimers. They then used both cell-based experiments and a mouse model to examine how these receptor pairs influence cancer.
Their results showed that dimerized VIPR2 contributes to breast tumor growth and metastasis through the region where the two receptor molecules connect. This interaction occurs at a portion of the receptor known as the transmembrane domains (TM)3 and 4. These regions can either strengthen the pairing between two VIPR2 molecules or prevent it from forming.
Whether the receptors remain paired depends on the presence of small chains of amino acids called TM3-4 peptides. Amino acids are the building blocks that make up proteins. The TM3-4 peptides interfere with the way the transmembrane domains interact with each other.
“We found that VIPR2 moved further apart in cells expressing TM3-4-peptides, suggesting that TM3-4 prevents VIPR2 dimerization,” Asano said. “Breast cancer cells stably expressing TM3-4 region exhibited suppressed tumor growth and lymph-node metastasis.”
This process of breaking apart dimers is called de-dimerization.
“VIPR2 de-dimerized by expression of TM3-4 had reduced affinity for specific proteins and was no longer involved in the activation of signaling pathways involved in cell proliferation and metastasis,” said co-corresponding author Yukio Ago, professor in the Department of Cellular and Molecular Pharmacology at Hiroshima University’s Graduate School of Biomedical and Health Sciences. “Since the expression of TM3-4 was able to suppress the progression of breast cancer cells, we plan to verify the anticancer effect of the purified TM3-4 peptide in animal models. Our goal is to develop novel anticancer drugs that target cancer cells in which dimerization is enhanced due to increased expression of VIPR2.”
Reference: “Dimerisation of the VIP receptor VIPR2 is essential to its binding VIP and Gαi proteins, and to its functions in breast cancer cells” by Satoshi Asano, Kairi Ozasa, Teru Uehara, Rei Yokoyama, Takanobu Nakazawa, Souichi Yanamoto and Yukio Ago, 9 April 2025, British Journal of Pharmacology.
DOI: 10.1111/bph.70039
The Japan Society for the Promotion of Science, the Japan Agency for Medical Research and Development, the Hiroshima University Fund “Nozomi H Foundation” and the Tokyo Biochemical Research Foundation supported this research.
This paper received funding from Hiroshima University to cover open access fees.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Breast Cancer molecules-
Is there specific localisation of VIPR2 I.e specific tissue membrane localisation and time related upregulation, dynamic or static. What is the size of molecules and how are they going to manipulated?