
Brazilian researchers found that boosting the synthesis of hevin, a glycoprotein naturally secreted by astrocytes, led to enhanced neuronal connectivity in rodents.
In Brazil, researchers from the Federal University of Rio de Janeiro (UFRJ), along with collaborators from the University of São Paulo (USP), have discovered a molecule called hevin that can reverse cognitive impairment. The study, conducted in mice, revealed that this glycoprotein, produced by brain cells known as astrocytes, can increase the connections between neurons (synapses) in both aged mice and in animal models of Alzheimer’s disease.
“Hevin is a well-known molecule involved in neural plasticity. It’s naturally secreted by cells in the central nervous system that support the functioning of neurons and are known as astrocytes. We found that the overproduction of hevin is capable of reversing cognitive deficits in aged animals by improving the quality of synapses in these rodents,” says Flávia Alcantara Gomes, head of the Cellular Neurobiology Laboratory the UFRJ Institute of Biomedical Sciences.
The study, published in the journal Aging Cell, was supported by the Ministry of Health, the Carlos Chagas Filho Foundation for Research Support in the State of Rio de Janeiro (FAPERJ), and FAPESP.
It’s important to note that there is still a long way to go before a molecule involved in reversing cognitive impairment can become a drug. This is a basic science study in mice. Another key consideration is whether the compound can cross the blood-brain barrier, which would require efforts to design molecules with this property and the same therapeutic potential.
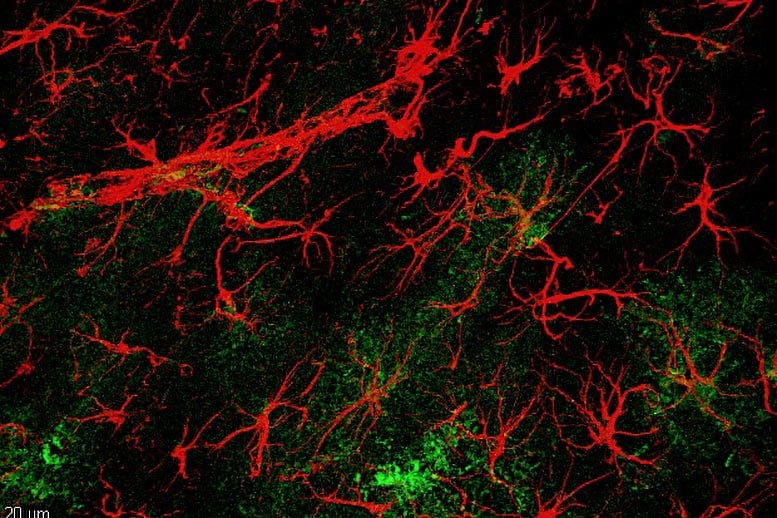
Rodent hippocampal astrocytes (in red) overexpressing hevin (in green). Credit: Felipe Cabral-Miranda and Ana Paula Bergamo Araujo“Of course, in the future it’ll be possible to develop drugs that have the same effect as hevin. For now, however, the fundamental benefit of this work is a deeper understanding of the cellular and molecular mechanisms of Alzheimer’s disease and the aging process. The originality lies in understanding the role of the astrocyte in this process. We’ve taken the focus away from neurons, shedding light on the role of astrocytes, which we’ve shown could also be a target for new treatment strategies for Alzheimer’s disease and cognitive impairment,” says Gomes.
Evidence-based hypothesis
By observing public data, the researchers found that hevin levels in the brain of Alzheimer’s patients decrease compared to healthy individuals of the same age. With this information and using a recombinant viral vector, the group at the UFRJ Institute of Biomedical Sciences overexpressed hevin in astrocytes from aged animals and in transgenic animal models of Alzheimer’s disease.
In addition, the set of proteins produced by the brain cells (brain proteome) of these animals was also analyzed. Comparing the animals with and without hevin overproduction, the researchers found that 89 proteins were differentially expressed. This stage of the work was carried out in the “Redox Proteomics Core” Multiuser Laboratory of the Center for Redox Processes in Biomedicine (Redoxoma) – a FAPESP Research, Innovation and Dissemination Center (RIDC) based at the University of São Paulo’s Institute of Chemistry (IQ-USP).
“The synapse depends on proteins to release a chemical signal from one neuron to another. Proteomic analysis showed that hevin reinforcement in astrocytes regulates different groups of proteins involved in synapses. We observed an increase in synapses, or in other words, a closer connection between neurons and, consequently, better cognitive performance,” explains Danilo Bilches Medinas, professor at the IQ-USP Department of Biochemistry.
Amyloid plaques
In addition to identifying the potential of hevin to reverse cognitive deficits in rodents through behavioral testing, the researchers also observed that overexpression of the molecule in astrocytes did not affect the deposition of beta-amyloid plaques in the hippocampus – a hallmark of Alzheimer’s disease that has been the focus of disease studies and a target for drug development.
“To our surprise, although the cognitive deficit was reversed in Alzheimer’s model animals, there was no change in the content of the plaques. This highlights the complexity of the disease in terms of having a multifactorial mechanism. This is illustrated by older people who have plaque formation but show no symptoms of the disease,” says Felipe Cabral-Miranda, a biomedical scientist at the UFRJ Institute of Biomedical Sciences and first author of the study.
“Although there’s still no consensus among researchers, I work with the hypothesis that the formation of beta-amyloid plaques isn’t the cause of Alzheimer’s. And the results of the study, by providing proof of concept for a molecule that can reverse cognitive decline without affecting beta-amyloid plaques, support the hypothesis that these, although involved in the mechanisms of the pathology, aren’t enough to cause Alzheimer’s,” he adds.
Reference: “Astrocytic Hevin/SPARCL-1 Regulates Cognitive Decline in Pathological and Normal Brain Aging” by Felipe Cabral-Miranda, Ana Paula Bergamo Araujo, Danilo Bilches Medinas and Flávia Carvalho Alcantara Gomes, 12 February 2025, Aging Cell.
DOI: 10.1111/acel.14493
The study was funded by the São Paulo Research Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
In the interest of perverse risk aversion, we need to make sure we exercise extreme caution so that this is only found to be perfectly safe after 40 years of studies. The fact that people die waiting for this make absolutely no difference to how we assess the risk. You just can’t be too safe.
Note 2506070545_Source1. Analyzing【
The havin molecule that fundamentally blocks the cognitive decline of Alzheimer’s is in the j.bank solution, which has been shown as junk dna. Uh-huh. For neuronal connections to be active, the cause of brainwashing, we need to know msbase.story. Huh. Maybe at the age of 100, the brain can get an Einstein class IQ of 300 with the brain of a young man. Uh-huh.
The number of Alzheimer’s patients worldwide is estimated to be around 55 million in 2019, and it is expected to increase to around 114 million by 2050. Alzheimer’s disease is the most common cause of dementia in the elderly. Around the world, 3.9% of the population aged 60 or older suffers from dementia, of which Alzheimer’s disease accounts for the majority.
The number of people with dementia is expected to increase:
The World Health Organization (WHO) predicts that the number of dementia patients worldwide will increase to 114 million by 2050.
_[2~3] Close connection between haze molecule neurons that increase synapses signals and improve cognitive function.
This indicates that in msbase.mode, the molecule that dictates the ordered connectivity within the nk protein is havin qpeoms. This restores the cognitive power of the ordered magic.roader connection between nk neurons. In example 1, it is seen as a plaque shown as dna.junk of repetitive n.qpeoms.bance(*) Hevin, but in reality, only n-n.banc=banc remains in n.banc, and we found the unique factor of havin qpeoms that helps neuron connections. Uh-huh.
The magnetum loader of neurons (=|=>)
View 1. (=|=>)
04110613=0banc.ms(b.ms)
14051203
15080902
01100716
=View 1.-9junk (.banc)
.vix.4.habin.plaque
.junk_dna.=|=>
00000004
05000000
00000000
00010000
04110604=-9b.ms
05051203
15080002
01010716
≈≈≈=========
https://scitechdaily.com/scientists-discover-natural-molecule-that-reverses-cognitive-decline/
1.
Scientists have discovered a natural molecule that reverses cognitive decline.
Brazilian researchers discovered that hebin, a brain protein, can reverse cognitive decline in mice, opening a new way for Alzheimer’s research by bypassing the long-targeted beta-amyloid plaque. Promoting the synthesis of hebin, a glycoprotein naturally secreted from astrocytes, improves rodent nerve connectivity.
[Found a molecule called hevin that can reverse cognitive impairment].
The study, conducted on mice, showed that this glycoprotein, produced by brain cells known as astrocytes, can increase the connection between neurons (synapses) in both aging mice and Alzheimer’s disease animal models.
1-1.
Hevin is a well-known molecule associated with neuroplasticity. It is naturally secreted from astrocytes that support neuron function in the central nervous system. It has been found that excessive production of hevin can reverse cognitive decline in older animals by improving synaptic quality in rodents.
1-2.
It should be noted that there is still a long way to go before the molecules involved in reversing cognitive impairment are developed into drugs. This is a basic scientific study of mice. Another important consideration is whether this compound can cross the blood-brain barrier, which requires efforts to design molecules with these properties and the same therapeutic potential.
1-3.
Of course, in the future, drugs with the same effects as hevin could be developed. However, for now, the fundamental benefit of this study is to gain a deeper understanding of the cellular and molecular mechanisms of Alzheimer’s disease and the aging process. Understanding the role of astrocytes in this process is originality. We focused on the role of astrocytes away from nerve cells, which we have shown could be the target of new therapeutic strategies for Alzheimer’s disease and cognitive impairment.
2. Evidence-based hypothesis
The researchers observed public data and found that hevin levels in the brains of Alzheimer’s patients decreased compared to healthy people of the same age group.
2-1.
Using this information and recombinant viral vectors, the UFRJ Biomedical Laboratory team overexpressed hevin in astrocytes and Alzheimer’s disease transgenic animal models in elderly animals.
2-2.
In addition, we also analyzed a set of proteins (brain proteomes) produced by brain cells in these animals. The researchers compared animals with and without hebin overproduction and found that 89 proteins were expressed differently.
2-3.
Synapses depend on proteins that emit chemical signals from one neuron to another. Proteomic analysis showed that hebin enhancement of astrocytes regulates various protein groups involved in synapses. It has been observed that increased synapses, that is, connections between neurons become closer and, consequently, cognitive function is improved.
3. Amyloid plaque
The researchers not only identified Hevin’s potential to reverse cognitive defects in rodents through behavioral tests, but also observed that overexpression of this molecule in astrocytes does not affect the accumulation of beta-amyloid plaques in the hippocampus. Beta-amyloid plaques are characteristic of Alzheimer’s disease, and are a focus of disease research and a target of drug development.
Surprisingly, cognitive impairment recovered in Alzheimer’s disease model animals, but there was no change in plaque content. This emphasizes the complexity of the disease in that it has a multifactorial mechanism. This can be confirmed in elderly people who have formed plaques but do not show symptoms of the disease.
3-1.
Although there is no consensus among researchers yet, I am studying with the hypothesis that beta-amyloid plaque formation is not the cause of Alzheimer’s disease.