
A newly identified cancer pathway may finally reveal how to stop pancreatic tumors before they spread.
A Cornell-led research effort has uncovered how a highly aggressive type of pancreatic cancer is able to enter the bloodstream, addressing a long-standing question about how the disease spreads and pointing to a potential therapeutic target.
Pancreatic ductal adenocarcinoma is one of the deadliest cancers, with less than 10% of patients surviving for five years after diagnosis. The tumor is surrounded by a thick, fibrotic microenvironment that functions like protective armor. This dense tissue complicates drug delivery and should, in principle, limit the tumor’s ability to spread. Despite this, the cancer spreads readily, a contradiction that has challenged researchers for years.
ALK7 identified as a dual driver of metastasis
Findings published in Molecular Cancer show that a receptor known as ALK7 plays a central role by triggering two linked biological pathways. One pathway enables cancer cells to move more easily through a process called epithelial-mesenchymal transition, while the other produces enzymes that degrade the walls of blood vessels.
“In other words, ALK7 gives pancreatic cancer cells both the engine to move and the tools to invade,” said Esak Lee, co-corresponding author of the study and assistant professor in the Meinig School of Biomedical Engineering in Cornell Engineering. The co-first author is Anna Kolarzyk, Ph.D. ’25, postdoctoral researcher.
The team’s results help clarify previous studies that had shown conflicting roles for ALK7, with some suggesting it slowed metastasis and others indicating it promoted it. By using mouse models and organ-on-chip platforms that replicate human blood vessels, the researchers demonstrated that inhibiting ALK7 greatly reduced the cancer’s ability to metastasize.
Organ-on-chip models reveal the timing of cancer invasion
The organ-on-chip system, developed in Lee’s lab, simulates the tumor microenvironment and is superior to animal models for studying different stages of the cancer. Using it, the researchers studied whether ALK7 drives the initial invasion of blood vessels or the later stage, when circulating tumor cells exit the bloodstream to form new tumors in organs such as the lungs or liver. What they found was that cancer cells couldn’t enter blood vessels when ALK7 was inhibited. But when they mimicked a later stage of cancer by placing the cells inside the vessels, they spread quickly, indicating that the timing for treatment is crucial.
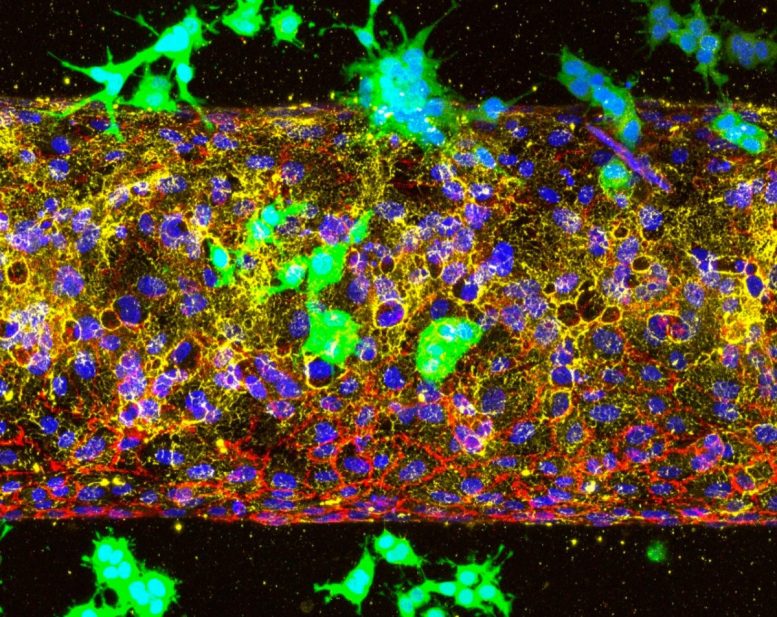
“Once we miss this early opportunity to block ALK7 receptors, the cancer cells can freely circulate in the bloodstream and easily seed into other organs,” Lee said. “But if we can inhibit ALK7 at the cancer’s earliest and most vulnerable stage, we might see better outcomes for patients.”
Broader implications for future cancer research
The study also highlights the potential to apply organ-on-chip platforms to study other types of cancers, or how immune cells infiltrate and exit vessels.
“Some cancers have very different microenvironments, so, potentially, ALK7 might show different impacts,” Lee said. “I hope this study really opens a new avenue for cancer research.”
Reference: “Non-canonical ALK7 pathways promote pancreatic cancer metastasis through β-catenin/MMP-mediated basement membrane breakdown and intravasation” by Anna M. Kolarzyk, Yujin Kwon, Elizabeth Oh, Keng-Jung Lee, Su-Yeon Cho, Issahy Cano, Renhao Lu, Tae Joon Kwak, Jaehyun Lee, Gigi Wong, Andrew H. Kim, Omar Gandarilla, Manuel Hidalgo, Won Kyu Kim and Esak Lee, 4 July 2025, Molecular Cancer.
DOI: 10.1186/s12943-025-02384-w
The research was supported by the National Institutes of Health, the National Science Foundation and the National Research Foundation of Korea.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.