Johns Hopkins scientists discovered a way to convert “immune-cold” tumors into “immune-hot” ones by activating key immune pathways.
In studies using mouse models of breast, pancreatic, and muscle cancers, scientists at Johns Hopkins All Children’s Hospital have uncovered new evidence that a unique approach to stimulating the body’s natural immune defenses can help prevent cancer from returning and improve survival rates.
The research, published in Nature Immunology, received federal support from the National Cancer Institute/NIH.
Cancerous tumors are often labeled “immune cold” or immune-suppressive because the immune system fails to recognize them as threats. As a result, these tumors resist attack from immune cells and respond poorly to standard treatments, leading to less favorable outcomes for patients.
The Johns Hopkins team set out to find a way to transform these immune-cold tumors into “immune hot” ones—tumors that the immune system can detect and target. By doing so, they aimed to make immune cells such as B cells and T cells more effective at destroying cancer cells and to improve how patients respond to both chemotherapy and immunotherapy.
Enhancing Tumor Immunity Through TLS Formation
Building on their earlier research in breast cancer, the research team hypothesized that “spicing up” the tumor environment with immune-activating agents improves the “fitness” of tertiary lymphoid structures (TLSs) and dramatically enhances immune responses to target tumors.
TLS are clusters of lymphocytes that form in areas of chronic inflammation, including immune-hot tumors. These structures are critical in helping the immune system fight cancer, and their presence strongly correlates with improved treatment responses and patient survival.
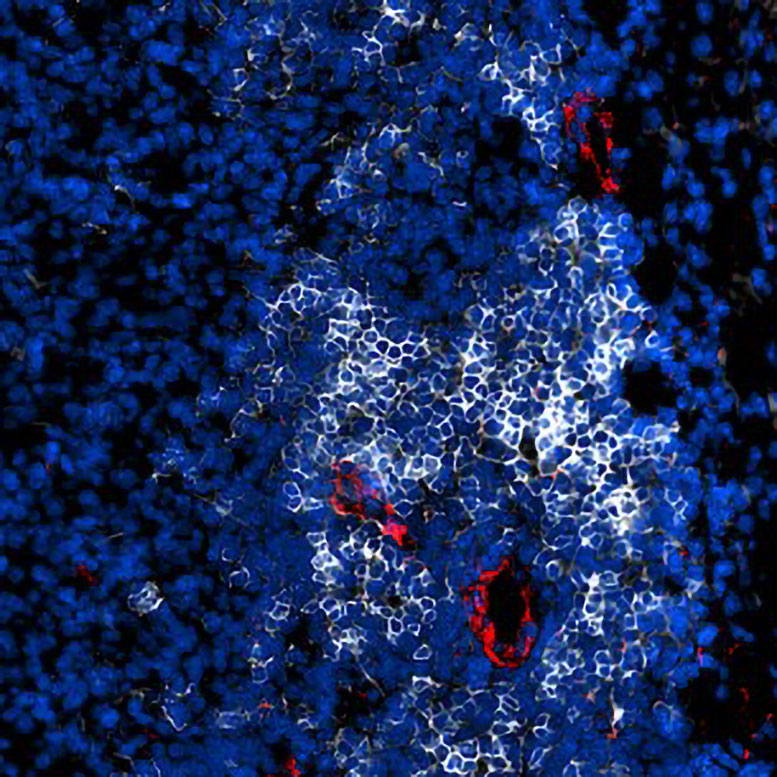
To test their approach, the researchers “reverse-engineered” a TLS-rich tumor environment to identify the stimuli required for TLS formation. They then applied these stimuli to TLS-free tumors growing in mice, administering two immune-activating substances (agonists) that stimulate the protein STING and the lymphotoxin-β receptor (LTβR).
Dual activation of STING and LTβR triggered a rapid response from killer T cells (CD8⁺ T cells), leading to strong tumor growth inhibition. The treatment also induced the formation of high endothelial venules, the specialized blood vessels that admit lymphocytes into tissues. These blood vessels functioned like dedicated gateways, enabling large numbers of T and B cells to enter the tumors and assemble into TLSs.
Establishing Long-Term Immune Protection
Within these TLS, B cells initiated germinal‑center reactions, matured into antibody‑secreting plasma cells, and generated long‑lived memory cells. Tumor‑specific IgG antibodies were detected, and plasma cells persisted in the bone marrow—evidence of durable, systemic immunity that can help protect against relapse.
Treatment also increased helper (CD4⁺) T cells and memory CD8⁺ T cells and balanced immune signaling, strengthening both antibody‑mediated (humoral) and cell‑mediated immunity.
Together, the researchers say, the findings suggest early and combined efforts to boost T‑cell activity not only kill tumor cells directly but also induce TLS maturation that sustains and amplifies anti-tumor responses.
“Our findings show that we can therapeutically induce functional TLS in otherwise immune‑cold tumors,” says Masanobu Komatsu, Ph.D., principal investigator of the study and senior scientist at the Johns Hopkins All Children’s Cancer & Blood Disorders Institute. “By building the right immune infrastructure inside tumors, we can potentiate the patient’s own defenses—both T cell and B cell arms—against cancer growth, relapse, and metastasis.”
Because TLS abundance correlates with better outcomes across many tumor types, the use of the two protein stimulators together may offer a broadly applicable way to enhance the effectiveness of existing therapies, including checkpoint inhibitors that are the mainstay of immunotherapies, and traditional chemotherapy.
Komatsu’s team is further investigating the mechanism of action of TLS therapy and preparing for its clinical application in adult and pediatric cancer patients.
Reference: “Simultaneous STING and lymphotoxin-β receptor activation induces B cell responses in tertiary lymphoid structures to potentiate antitumor immunity” by Junko Sawada, Yasuhiro Kikuchi, Maxwell Duah, Jose Luis Herrera, Fumiaki Kanamori, Krisztian Csomos, Tomoko Stansel, Nobuyoshi Hiraoka, Masayuki Yoshida, Jolan Walter, Carl F. Ware and Masanobu Komatsu, 2 September 2025, Nature Immunology.
DOI: 10.1038/s41590-025-02259-8
This research was supported by the National Cancer Institute/NIH R01 grants, the Department of Defense Congressionally Directed Cancer Research Program, and the Florida Department of Health Bankhead Coley Cancer Research Program.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
My husband has Pancreatic Cancer, our daughter passed away 3 yrs ago from it. He has his tumor removed in Jan of this year. His C9-19 mar k ers are in the LOWER 20’s. Can he be a candidate for this.
PLEASE HELP US!!!
DIANNE E FILLYAW
[email protected]
850-960-9006
https://scitechdaily.com/johns-hopkins-anti-parasitic-drug-slows-pancreatic-cancer-in-mice/
Search for Dr.Robert Morse videos
Dr.Robert Morse videos online