Optical Nanomotion Detection Revolutionizes Antibiotic Testing With Rapid, Low-Cost, Single-Cell Precision.
A new method for quick antibiotic susceptibility testing using optical microscopy has been created by scientists from EPFL and Vrije Universiteit Brussel. The approach, called Optical Nanomotion Detection, is fast, accurate at the single-cell level, label-free, and can be carried out using a basic optical microscope, equipped with a camera or a mobile phone.
“We have developed a technique in our laboratories that allows us to obtain an antibiogram within 2-4 hours – instead of the current 24 hours for the most common germs and one month for tuberculosis,” says Dr. Sandor Kasas at EPFL. Professor Ronnie Willaert at Vrije Universiteit Brussel adds: “Our technique is not only faster but also simpler and much cheaper than all those existing now.”
Antibiotic resistance occurs when bacteria gain the capability to overcome the medicines created to eradicate them. It has now become a worldwide concern for public health. In 2019, it was the cause of at least 1.27 million fatalities globally and was implicated in nearly five million deaths. In the United States, there are nearly three million cases of antimicrobial-resistant infections annually, and the cost of treating the top six of these infections exceeds $4.6 billion. In the European Union, there are nearly 700,000 cases each year, with an estimated cost of €1.5 billion.
Antibiotic sensitivity testing (AST) uses culture methods that expose bacteria to antibiotics, or genetic methods to determine if bacteria possess genes that confer resistance. Typical ASTs last up to 24 hours or even longer for slow-growing bacteria – a timeframe that can mean life or death in a clinical setting. There have been some faster ASTs developed in recent years, but they tend to be complex, needing sophisticated and expensive equipment.
Revolutionizing Testing with Optical Nanomotion Detection
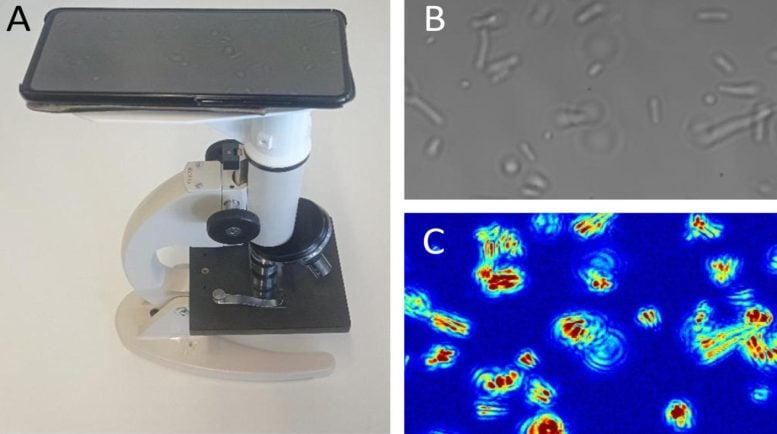
Now, researchers led by Kasas and Willaert have developed a fast, cheap, and widely accessible method based on optical microscopy that can perform an AST with single-cell sensitivity without needing to attach or label bacteria. The technique uses a basic, conventional optical microscope, a camera or mobile phone, and dedicated software. The joint research project was published in the journal PNAS.
The new technique is called optical nanomotion detection (ONMD), and involves the monitoring of nanoscale vibrations of single bacterial before and while being exposed to antibiotics. The monitoring is performed with a basic optical microscope, a video camera, or a mobile phone.
The ONMD technique monitors the microscopic oscillations of bacterial cells (nanomotion) that characterize living organisms and can be considered as a “signature of life.” Indeed, nanomotion lasts as long as the organism is alive but stops immediately when it is dead. In the ONMD technique, bacterial nanomotion is recorded in a movie in which all individual cell displacements are monitored with sub-pixel resolution.
The researchers used ONMD to successfully detect the sensitivity of numerous bacteria to antibiotics. Escherichia coli, Staphylococcus aureus, Lactobacillus rhamnosus, and Mycobacterium smegmatis (a non-pathogenic bacterial model for tuberculosis) sensitivities to the antibiotics ampicillin, streptomycin, doxycycline, and vancomycin was determined in less than two hours.
The Game-Changing Potential of ONMD
The ONMD not only monitors the bacteria’s life-death transitions upon exposure to different antibiotics but also highlights changes in the bacteria’s metabolism caused by the availability of nutrients. The tests showed that ONMD can assess the sensitivity or resistance of bacterial cells to antibiotics in a simple and rapid way by monitoring cellular oscillations.
The authors state: “The simplicity and efficiency of the method make it a game-changer in the field of AST” as it can be applied to a wide range of bacteria, which has significant implications for clinical and research applications.
Reference: “Simple optical nanomotion method for single-bacterium viability and antibiotic response testing” by Maria I. Villalba, Eugenia Rossetti, Allan Bonvallat, Charlotte Yvanoff, Vjera Radonicic, Ronnie G. Willaert and Sandor Kasas, 24 April 2023, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2221284120
The study was funded by the Swiss National Science Foundation, Research Foundation – Flanders (FWO), the Belgian Federal Science Policy Office (Belspo), and the European Space Agency.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.