The display of a smartphone reacts to finger pressure. The carnivorous Venus flytrap, on the other hand, even notices when a lightweight like a fly lands on it. Special genes make this possible.
All plant cells can be made to react by touch or injury. The carnivorous Venus flytrap (Dionaea muscipula) has highly sensitive organs for this purpose: sensory hairs that register even the weakest mechanical stimuli, amplify them, and convert them into electrical signals that then spread quickly through the plant tissue.
Researchers from Julius-Maximilians-Universität (JMU) Würzburg in Bavaria, Germany, have isolated individual sensory hairs and analyzed the gene pool that is active in catching insects. “In the process, we found for the first time the genes that presumably serve throughout the plant kingdom to convert local mechanical stimuli into systemic signals,” says JMU plant researcher Professor Rainer Hedrich.
That’s a fine thing, because virtually nothing was known about mechano-receptors in plants until now. Hedrich’s team presents the results in the open-access journal PLOS Biology.
Sensory hairs convert touch into electricity
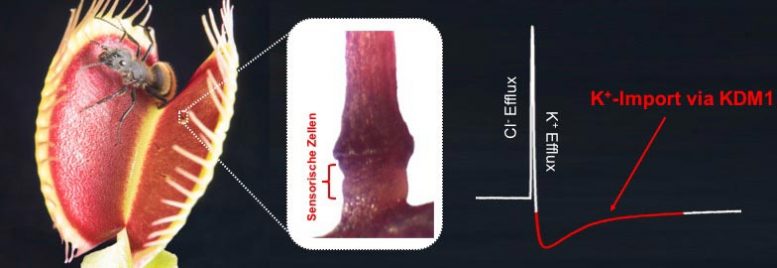
The hinged trap of Dionaea consists of two halves, each carrying three sensory hairs. When a hair is bent by touch, an electrical signal, an action potential, is generated at its base. At the base of the hair are cells in which ion channels burst open due to a stretching of their envelope membrane and become electrically conductive. The upper part of the sensory hair acts as a lever that amplifies the stimulus triggered by even the lightest prey.
These micro-force-touch sensors thus transform the mechanical stimulus into an electrical signal that spreads from the hair over the entire flap trap. After two action potentials, the trap snaps shut. Based on the number of action potentials triggered by the prey animal during its attempts to free itself, the carnivorous plant estimates whether the prey is big enough – whether it is worth setting the elaborate digestion in motion.
From genes to the function of the touch sensor
To investigate the molecular basis for this unique function, Hedrich’s team “harvested” about 1000 sensory hairs. Together with JMU bioinformatician Professor Jörg Schultz, they set out to identify the genes in the hairs.
“In the process, we noticed that the fingerprint of the genes active in the hair differs from that of the other cell types in the trap,” says Schulz. How is the mechanical stimulus converted into electricity? “To answer this, we focused on the ion channels that are expressed in the sensory hair or are found exclusively there,” says Hedrich.
In search of further ion channels
The sensory hair-specific potassium channel KDM1 stood out. Newly developed electrophysiological methods showed that without this channel, the electrical excitability of the sensory hairs is lost, i.e. they can no longer fire action potentials. “Now we need to identify and characterize the ion channels that play an important role in the early phases of the action potential,” Hedrich said.
Reference: “The Venus flytrap trigger hair–specific potassium channel KDM1 can reestablish the K+ gradient required for hapto-electric signaling” by Anda L. Iosip, Jennifer Böhm, Sönke Scherzer, Khaled A. S. Al-Rasheid, Ingo Dreyer, Jörg Schultz, Dirk Becker, Ines Kreuzer and Rainer Hedrich, 9 December 2020, PLOS Biology.
DOI: 10.1371/journal.pbio.3000964
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Very Interesting Research.
Venus Flytrap has alwyas fascinated me.
I see it is as the missing link between Carnivorours Plant Species and Regular Plant Species.
Sharing Some Thoughts which may seem far fetched.
((1) 2.9 degrees deflection and 3 mg. mosquito will spring the trap. Hypersensitive. More sensitive when young. Less when Adult. Nutrient requirements may drive the speed of trigger.
(2) Interesting observation at Constriction site where bending hair trigger translates into Shear Stress. Connect with Shear Stress which causes earthquakes when continental plates scrape against each other.Linking Micro with Macro.
(3) Single layered mechanoreceptor drive mechanotransdction. Link with Quantum Chemistry. Single Layer Graphite and quantum effects. Link with Human Cells. Digestive and Neuro biology systems. Mechanical to electrical energy. Nano Chemistry.
(4) Why missing Link? Plant with Palsmodesmata Channels and Ion Channels which are highly specific and action potential in 4 out of 5 triggers.Plant attracts and traps insects and digests them. Action Potential can also be elicited by compressing the fly-traps non-trigger tissue by a force more than 1000 times greater than the hypersensitive Trigger invented by Natue.How to make hypersensitive trigger mechanism clues. Copy nature.
From a Genetic Perspective, we will have much greater understanding after we complete the mapping of the Gene of All creatures and Plants and other life which coud theoritically evolve and also all which existed in the Past . I REPEAT All Life including the Human Genome , which is at around 8% currently.
Also the interaction of the environment of all inanimate and animate materials which once they enter the Human Being can cause disease , failure of Organs and death.
We have barely got the knowledge of this area of Science. The function of ALL the Nucleotides and the impact of interactions of various disease causing pathogens, viruses and Bacteria and which ones could cause the extinction of which species of plants and animals and over what timeframe could be better understood.
I think medical professionals would agree that Prevention is FAR better than Cure. Identify and address the enemy within. Also, address the risk of the planet extinction of all life events from the events which are off-planet .Earth.
These are my views from a risk management perspective. Views expressed are personal and not binding on anyone.
May be helpful in keeping the Extinction whirlwind we have let loose on the Planet earth thanks to our disrespect for all life.
The brainpower of ALL Humanity needs to be brought on this if we are to avoid the fate of the Dodo.
ALL Streams of Knowledge and sharing the same seamlessly with All humans may be required to progress rapidly and find answers.