Supercomputer simulations reveal how RNA stabilizes Ebola’s structure, offering promising targets for antiviral drugs that could halt the virus by locking it in place.
In the midst of a global pandemic with COVID-19, it’s hard to appreciate how lucky those outside of Africa have been to avoid the deadly Ebola virus disease. It incapacitates its victims soon after infection with massive vomiting or diarrhea, leading to death from fluid loss in about 50 percent of the afflicted. The Ebola virus transmits only through bodily fluids, marking a key difference from the COVID-19 virus and one that has helped contain Ebola’s spread.
Unraveling Ebola’s Genetic Armor
Ebola outbreaks continue to flare up in West Africa, although a vaccine developed in December 2019 and improvements in care and containment have helped keep Ebola in check. Supercomputer simulations by a University of Delaware team that included an undergraduate supported by the XSEDE EMPOWER program are adding to the mix and helping to crack the defenses of Ebola’s coiled genetic material. This new research could help lead to breakthroughs in treatment and improved vaccines for Ebola and other deadly viral diseases such as COVID-19.
“Our main findings are related to the stability of the Ebola nucleocapsid,” said Juan R. Perilla, an assistant professor in the Department of Chemistry and Biochemistry at the University of Delaware. Perilla co-authored a study published in October 2020 in the AIP Journal of Chemistry Physics. It focused on the nucleocapsid, a protein shell that protects against the body’s defenses the genetic material Ebola uses to replicate itself.
“What we’ve found is that the Ebola virus has evolved to regulate the stability of the nucleocapsid by forming electrostatic interactions with its RNA, its genetic material,” Perilla said. “There is an interplay between the RNA and the nucleocapsid that keeps it together.”
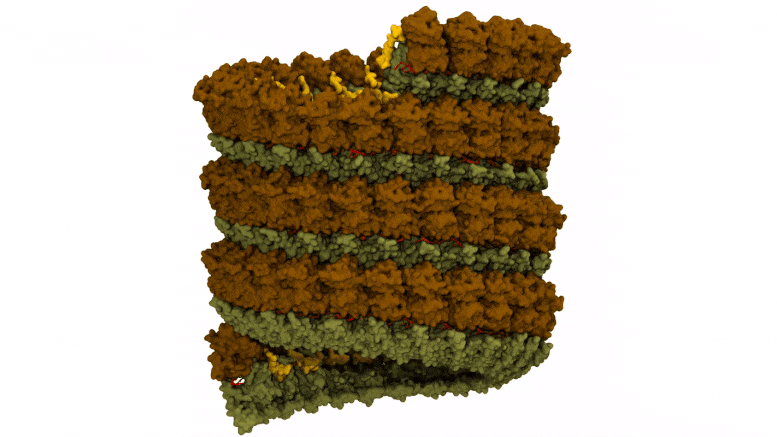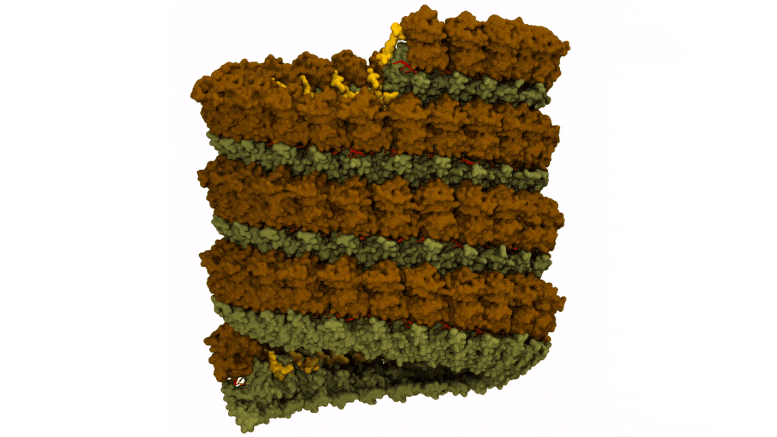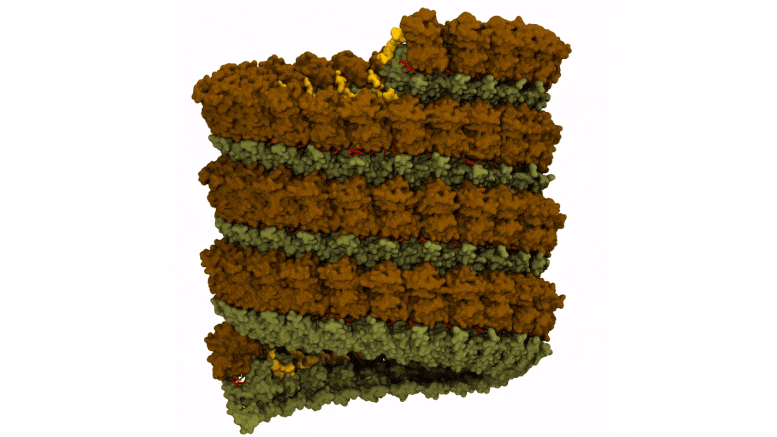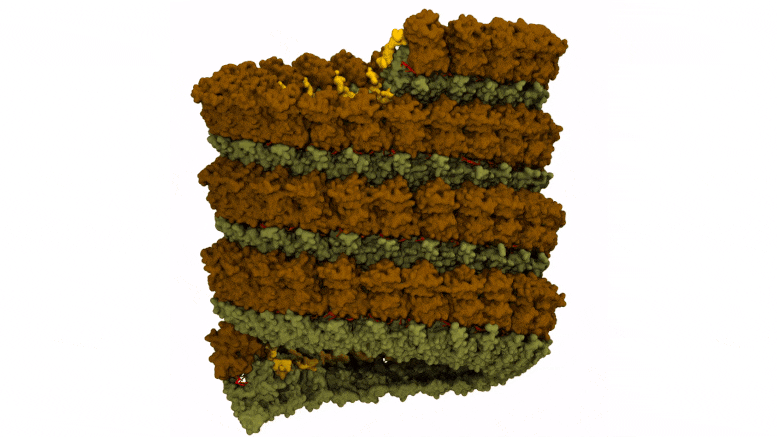
Like coronaviruses, the Ebola virus depends on a rod-like and helically-shaped nucleocapsid to complete its life cycle. In particular, structural proteins called nucleoproteins assemble in a helical arrangement to encapsulate the single-stranded viral RNA genome (ssRNA) that forms the nucleocapsid.
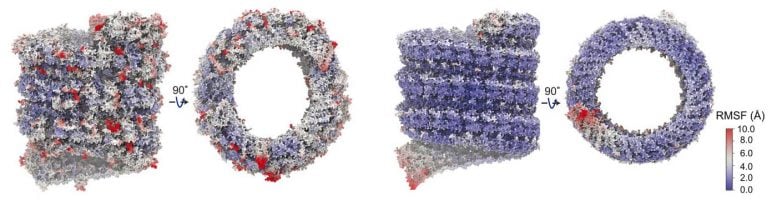
Preparation of Ebola virus nucleocapsid systems for atomistic molecular dynamics simulations. The virus monomer contained three nucleoprotein structural domains: N-terminal arm (yellow), N-terminal lobe (brown), and C-terminal lobe (dark green), and a bound RNA segment (red). Credit: Juan R. Perilla, University of Delaware
The study by Perilla and his science team sought the molecular determinants of the nucleocapsid stability, such as how the ssRNA genetic material is packaged, the electrostatic potential of the system, and the residue arrangement in the helical assembly. This knowledge is essential for developing new therapeutics against Ebola. Yet these insights remain out of reach even by the world’s best experimental labs. Computer simulations, however, can and did fill that gap.
“You can think of simulation work as a theoretical extension of experimental work,” said study co-author Tanya Nesterova, an undergraduate researcher in the Perilla Lab. “We found that RNA is highly negatively charged and helps stabilize the nucleocapsid through electrostatic interaction with the mostly positively charged nucleoproteins,” she said.
Nesterova was awarded funding through an XSEDE Expert Mentoring Producing Opportunities for Work, Education, and Research (EMPOWER) scholarship in 2019, which supports undergrads participate in the actual work of XSEDE.
“It was an effective program,” she said. “We used computational resources such as Bridges this summer. We also had regular communication with the coordinator to keep our progress on track.”
Molecular surface representation of the Ebola virus nucleocapsid with bound RNA. Credit: Juan R. Perilla, University of Delaware
Building the Virtual Ebola Model
The team developed a molecular dynamics simulation of the Ebola nucleocapsid, a system that contains 4.8 million atoms. They used the cryo-electron microscopy structure of the Ebola virus published in Nature in October of 2018 for their data in building the model.
“We built two systems,” said study co-author Chaoyi Xu, a PhD student in the Perilla Lab. “One system is the Ebola nucleocapsid with the RNA. And the other one is just the nucleocapsid as a control.”
“After we built the whole tube, we put each nucleocapsid in an environment that is similar to the cell,” Xu explained. They basically added sodium chloride ions, and then adjusted the concentration to match that found in the cytoplasm. They also put a water box inside around the nucleocapsid. “And then we ran a very powerful simulation,” Xu added.
The NSF-funded Extreme Science and Engineering Discovery Environment (XSEDE) awarded the team supercomputing allocations on the Stampede2 system at the Texas Advanced Computing Center and the Bridges system of the Pittsburgh Supercomputing Center.
“We are very thankful for the supercomputer resources provided by XSEDE that allowed this work to be possible. XSEDE also provided training through online courses that was helpful,” Xu said.

“On Stampede2, we have access to run simulations on hundreds or even thousands of nodes,” Xu continued. “This makes it possible for us to run simulations of larger systems, for example, the Ebola nucleocapsid. This simulation is impossible to finish locally. That’s very important,” he said.
“I like how with Bridges, when you run a simulation, you can be up to date on when it completes and when it started,” Nesterova added. She said that was helpful for creating Slurm scripts, which help manage and schedule jobs on compute clusters.
“We just started using Frontera for the Ebola project,” Xu added. Frontera is the NSF flagship Tier 1 system at TACC, ranked #9 in the world by Top500. “It’s more powerful because it has the latest CPU architecture. And it’s very fast,” he said.
“Frontera is part of the TACC infrastructure,” Perilla said. “We knew what developmental tools were going to be there, and also the queueing system and other intricacies of these machines. That helped a lot. In terms of architecture, we’re familiar with Stampede2, although this is a different machine. Our experience with Stampede2 allowed us to move quickly to start using Frontera,” he said.
The science team simulated the interaction of the atoms in the Ebola virus nucleocapsid and measured how they change in time, yielding useful information about the atomic interactions. One of the things they found was that without the RNA, the Ebola virus nucleocapsid kept its tube-like shape. But the packing of the nucleoprotein monomers became disordered, and its helical symmetry was lost. With the RNA, it kept its helix. Their results showed that the RNA binding stabilized the helix and preserved the structure of the Ebola virus nucleocapsid.
Pinpointing Molecular Interactions for Drug Design
The team also found important interactions between the nucleoprotein residues and the ssRNA, and also interactions between two nucleoproteins.
“There are two kinds of interfaces between the pairs of nucleoproteins that form the helical arrangement. We figured out which of these interfaces plays a more important role. We can either target that interface to destabilize the helical arrangement or stabilize the helical arrangement to a large extent such that the virus nucleocapsid is unable to disassemble,” said study co-author Nidhi Katyal, a postdoctoral researcher in the Perilla Lab.
The Ebola virus is one tough organism because it tightly regulates its macromolecular assembly. Perilla suggested that instead of trying to devise drugs that destroy the nucleocapsid, a good strategy might be to do the opposite.
“If you make it too stable, that’s enough to kill the virus,” he said. Borrowing a strategy from his background in HIV research, he wants to find targets for drugs to over-stabilize the Ebola virus and keep it from releasing its genetic material, a key step in its replication.
A Future Framework for Fighting New Pathogens
Perilla suggested a similar strategy for other pathogens that are tightly regulated, such as coronaviruses and hepatitis B viruses. “They’re a sweet spot, so to speak. We know what confers stability. Other teams can look to see if maybe this is a good druggable site for making it hypostable or making it hyperstable,” Perilla said.
Looking ahead, Perilla indicated his lab will be looking more closely at the specifics of ssRNA sequence and whether it confers stability to the Ebola virus nucleocapsid tube. If it does, then some regions might be exposed and might be transcribed first, similar to what happens in the nucleus of the cell. Perilla said it would be “unheard of in a virus,” and extremely advanced behavior in terms of the RNA regulating transcription.
Said Perilla: “We know that there will be more pathogens that just keep coming, particularly with coronaviruses now, and they can stop the world. It’s beneficial to society having the ability to study not only one virus, but taking these techniques to study a new virus, something like coronaviruses. In addition, the ability to train new students, like Tanya, provides the taxpayers their money’s worth in terms of training the next generation, transferring knowledge from other viruses, and fighting the current problems.”
The study, “Molecular determinants of Ebola nucleocapsid stability from molecular dynamics simulations,” was published in the AIP Journal of Chemical Physics, October 2020. The co-authors are Chaoyi Xu, Nidhi Katyal, Tanya Nesterova, and Juan R. Perilla, Department of Chemistry and Biochemistry, University of Delaware. Study funding came from National Science Foundation, the Delaware Established Program to Stimulate Competitive Research (EPSCoR), and the US National Institutes of Health.
Reference: “Molecular determinants of Ebola nucleocapsid stability from molecular dynamics simulations” by Chaoyi Xu, Nidhi Katyal, Tanya Nesterova and Juan R. Perilla, 20 October 2020, Journal of Chemical Physics.
DOI: 10.1063/5.0021491
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.