Researchers have identified a promising new strategy to treat autoimmune diseases by targeting a specific molecular interaction within immune cells.
Scripps Research scientists have created a new group of drug compounds that reduce harmful inflammation without weakening the body’s ability to fight infections, a major challenge in treating autoimmune diseases. These compounds, known as ENDOtollins, act by disrupting a “molecular handshake” between two proteins inside immune cells.
The findings, published in Nature Chemical Biology, point to more precise treatment options for diseases such as lupus, rheumatoid arthritis, and juvenile arthritis, which together affect more than 15 million Americans.
“A key component of our approach is to begin by understanding the biological mechanisms at play,” says Sergio D. Catz, professor at Scripps Research and senior author. “By accomplishing this first, we can more easily target the pathway driving inflammation without affecting other important processes.”
Many current treatments for autoimmune disorders, including hydroxychloroquine, work by broadly blocking endosomes. These are compartments inside cells that sort and process incoming material, including molecules that trigger immune responses. While this strategy can be effective, it often causes side effects such as gastrointestinal issues and, in some cases, vision damage. These problems lead many patients to stop treatment.
Instead of taking a broad approach, Catz and his team focused on two proteins, Munc13-4 and syntaxin 7. These proteins must bind together for Toll-like receptors (TLRs), which are immune sensors, to activate within endosomes. This “molecular handshake” helps the immune system detect foreign DNA and RNA from viruses and bacteria.
In autoimmune diseases, however, TLRs can become overactive. They may mistakenly respond to the body’s own nucleic acids, such as those released from neutrophil-extracellular traps, leading to persistent and damaging inflammation even when no real threat is present.
Discovering ENDOtollins
Working with co-author Hugh Rosen, a professor at Scripps Research and the Pearson Family Chair, the team screened about 32,000 compounds with help from the institute’s Molecular Screening Center. They identified molecules that block the interaction between Munc13-4 and syntaxin 7 without interfering with other cellular processes. Since Munc13-4 is primarily found in immune cells, this approach allows for more targeted control of inflammation.
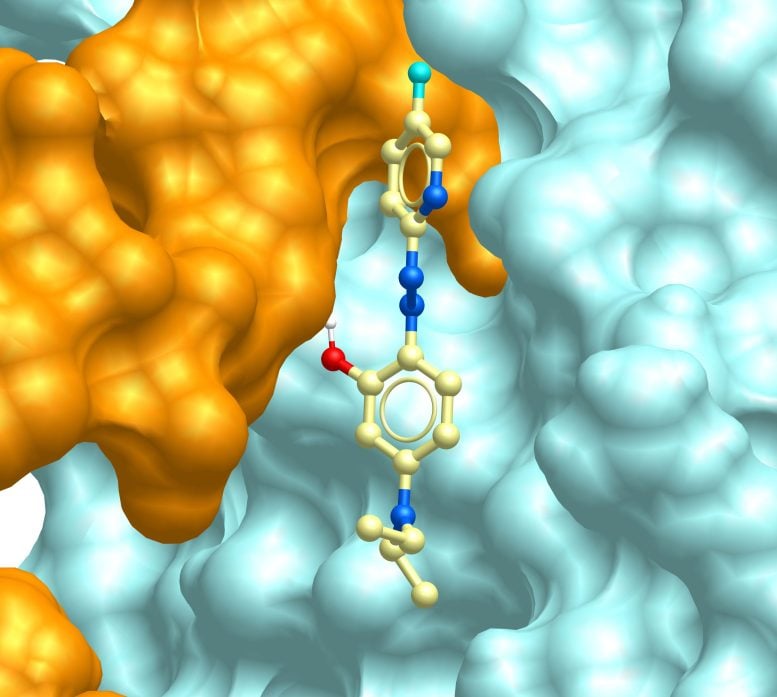
“Most treatments for autoimmune diseases manage symptoms; they don’t change the underlying course of the disease,” says Rosen. “What’s exciting about this approach is its potential to be disease-modifying: targeting the specific molecular machinery that drives inflammation, rather than broadly suppressing the immune system.”
A major advance in this work was the decision to screen compounds within intact cells. Many traditional methods remove proteins from their natural environment, but Catz and his team kept cellular compartments intact during testing.
“By maintaining the proteins in their natural environment, we increase the likelihood that compounds we find will actually work in living cells,” says Jennifer Johnson, first author and senior staff scientist at Scripps Research.
Promising Results in Models
The strongest candidate, ENDO12, lowered inflammation in animal models that were also exposed to a TLR-activating molecule. Treated animals showed a significant drop in inflammatory markers in the blood, including IL-6, IFN-γ, and the enzyme myeloperoxidase.
Importantly, ENDO12 did not reduce the animals’ ability to fight a real viral infection. They produced a normal antiviral immune response when exposed to a virus. This level of selectivity addresses a key concern with many immunosuppressive drugs, which can leave patients more vulnerable to infections.
The researchers plan to test ENDOtollins in models that better reflect human autoimmune diseases and to refine the compounds for possible clinical use.
Beyond autoimmune disorders, ENDOtollins may also help control cytokine storms. These are severe immune reactions seen in conditions such as advanced COVID-19 and as a side effect of CAR-T cancer therapy, both of which involve excessive IL-6 and uncontrolled inflammation.
Although clinical applications are still in the future, Catz notes that the underlying discoveries are valuable on their own. ENDOtollins could be used as precise tools to study other cellular processes linked to endosomes and lysosomes, including those involved in neurodegeneration and immune dysfunction. Gaining insight into how these compartments become stressed or fail could open new paths for understanding and treating a wide range of diseases.
Reference: “Munc13-4–STX7 inhibitors impair endosomal TLR activation and systemic inflammation” by Jennifer L. Johnson, Elsa Meneses-Salas, Aparna Shukla, Binchu Shaji, Farhana Rahman, Jing He, Evripidis Gavathiotis, Steve Brown, Rosana Gonzalez-Quintial, Hal M. Hoffman, Kristi L. Marquardt, John Teijaro, Catherine C. Hedrick, Roberto Baccala, Hugh Rosen and Sergio D. Catz, 6 April 2026, Nature Chemical Biology.
DOI: 10.1038/s41589-026-02181-6
This research was supported by U.S. Public Health Service grants R01HL088256, R01AR070837, P01HL152958 and R01DK110162, and by Cystinosis Research Foundation fellowships.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Dora the explorer catch the 🌟
thanks for this