
Scripps Research and Calibr scientists collaborated to discover potential therapeutic treatments for metabolic diseases.
“Metabolism” describes the body’s chemical changes that create the necessary ingredients for growth and overall health. Metabolites are the substances made and used during these metabolic processes—or, as a new discovery out of Scripps Research and its drug development arm, Calibr, indicates, they could also be potent molecules for treating severe diseases.
Researchers used cutting-edge drug discovery technologies to uncover a metabolite that converts white fat cells (“bad” fat) to brown fat (“good” fat) cells. This revelation offers a potential way of addressing obesity, type 2 diabetes, cardiovascular disease, and other metabolic conditions. Moreover, it speaks to the promise of using this creative drug discovery method to identify countless other potential therapeutics. The study was recently published in the journal Metabolites.
“The reason many types of molecules don’t go to market is because of toxicity,” says co-senior author Gary Siuzdak, PhD. “With our technology, we can pull out endogenous metabolites—meaning the ones that the body makes on its own—that can have the same impact as a drug with less side effects. The potential of this approach is even evidenced by the FDA’s recent approval of Relyvrio, the combination of two endogenous metabolites for the treatment of amyotrophic lateral sclerosis (ALS).” Siuzdak is the senior director of the Scripps Center for Metabolomics and professor of Chemistry, Molecular and Computational Biology at Scripps Research.
Brown Fat vs. White Fat: Key to Treating Metabolic Diseases
Metabolic diseases are often caused by an imbalance in energy homeostasis—in other words, when the body takes in more energy than it expends. This is why certain therapeutic approaches have centered around converting white fat cells (known as adipocytes) into brown fat cells. White adipocytes store excess energy and can eventually result in metabolic diseases like obesity, while brown adipocytes dissolve this stored energy into heat—ultimately increasing the body’s energy expenditure and helping bring it back into balance.
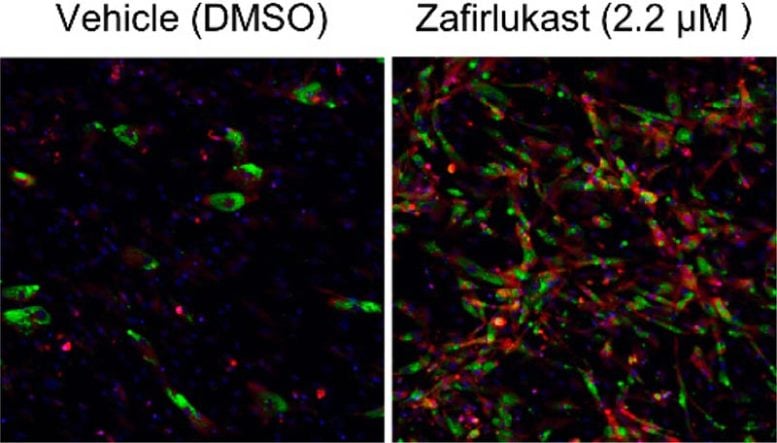
Identifying Zafirlukast and Its Role in Fat Cell Conversion
To uncover a therapy that could stimulate the production of brown adipocytes, the researchers searched through Calibr’s ReFRAME drug-repurposing collection—a library of 14,000 known drug compounds that have been approved by the FDA for other diseases or have been extensively tested for human safety. Using high-throughput screening—an automated drug discovery method for searching through large pools of information—the scientists scanned ReFRAME for a drug with these specific capabilities.
This is how they uncovered zafirlukast, an FDA-approved drug used for treating asthma. Through a set of cell culture experiments, they found zafirlukast could turn adipocyte precursor cells (known as preadipocytes) into predominantly brown adipocytes, as well as convert white adipocytes into brown adipocytes.
Myristoylglycine as a Non-Toxic Solution
While an encouraging find, zafirlukast is toxic when administered at higher doses, and it wasn’t entirely clear how zafirlukast was converting the fat cells. This is when the researchers partnered with Siuzdak and his team of metabolite experts.
“We needed to use additional tools to break down the chemicals in zafirlukast’s mechanism,” says Kristen Johnson, PhD, co-senior author of the paper and a director in Translational Drug Discovery Research at Calibr. “Framed another way, could we find a metabolite that was providing the same functional effect that zafirlukast was, but without the side effects?”
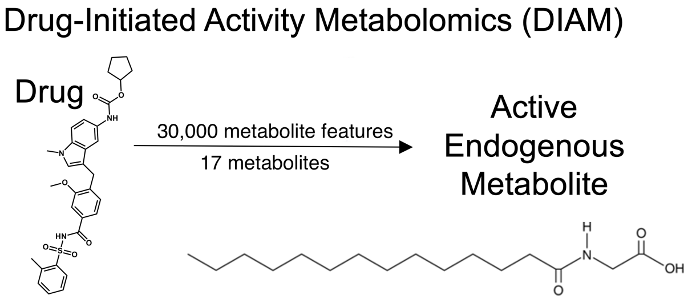
Siuzdak and his team designed a novel set of experiments, known as drug-initiated activity metabolomics (DIAM) screening, to help answer Johnson’s question. DIAM uses technologies such as liquid chromatography (a tool that separates components in a mixture) and mass spectrometry (an analytical technique that separates particles by weight and charge) to pool through thousands of molecules and identify specific metabolites. In this case, the researchers were searching through adipose tissue for metabolites that could lead to brown adipocyte cell production.
After reducing 30,000 metabolic features to just 17 metabolites, they found myristoylglycine—an endogenous metabolite that prompted the creation of brown adipocytes, without harming the cell. Of the thousands of metabolic features measured in the analysis, only myristoylglycine had this special characteristic, even among nearly structurally identical metabolites.
“Identifying myristoylglycine among the thousands of other molecules speaks to the power of Siuzdak’s approach and these technologies,” adds Johnson. “Our findings illustrate what happens when an analytical chemistry team and a drug discovery group closely collaborate with each other.”
Reference: “Drug-Initiated Activity Metabolomics Identifies Myristoylglycine as a Potent Endogenous Metabolite for Human Brown Fat Differentiation” by Carlos Guijas, Andrew To, J. Rafael Montenegro-Burke, Xavier Domingo-Almenara, Zaida Alipio-Gloria, Bernard P. Kok, Enrique Saez, Nicole H. Alvarez, Kristen A. Johnson and Gary Siuzdak, 16 August 2022, Metabolites.
DOI: 10.3390/metabo12080749
In addition to Siuzdak and Johnson, authors of the study, “Drug-Initiated Activity Metabolomics Identifies Myristoylglycine as a Potent Endogenous Metabolite for Human Brown Fat Differentiation” include Carolos Guijas, J. Rafael Montenegro-Burke, Xavier Domingo-Almenara, Bernard P. Kok and Enrique Saez of Scripps Research; and Andrew To, Zaida Alipio-Gloria and Nicole H. Alvarez of Calibr.
This research was partially funded by the National Institutes of Health and the NIH Cloud Credits Model Pilot.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
9 Comments
Human self-cheating with a destructive force: consume and process more energy to deposit as white adipose by spending resources like proteins/telomere length, convert it to brown tissue for wasting chemical heat = leaner body + shorter telomere. What a smart self-cheating/destruction: laziness glorified!
Shortening telomeres shortens mortality so it’s ironic and self defeating.
Enjoyed the artical. Is there more to it? Have you come up with a drug or concoction that will change the white to brown?
Take Montalukst every day for over 8 years & weigh 368 lbs. seem like it will work for weight loss.
Now you can just sell it to a pharmaceutical company to mark it up to an outrageous cost to the general public that needs it!
Yes, just take a pill. It will cure cancer. It will grow your hair. It will help you lose weight! Only wishful thinking
Want more brown fat and convert the bad fat to energy. Start taking cold showers and cold baths. Both have been proven to increase brown fat and eliminate the bad because it causes your body to increase it’s metabolism to fight the cold……
What’s that junk about telomeres in the comments? Grammar misses muddied it, and someone missed the title phrase. Just wow. Anyhow it’s cool enough to exercise now (unless you’re in Australia) so everyone good luck with the cryotherapy (shivering 10-20 min. every other day) and getting a good bit of exercise in to be in shape and warm again etc.
Understand, since I’m 74, probably not a good candidate for your testing. If you do consider me, that would be appreciated. Other than need to loose 25lbs, I’m in decent shape.