New research sheds light on the molecular processes determining the responsiveness of certain cancers to immunotherapy.
A new study has shed light on why immunotherapy does not always work in certain types of cancer. Led by researchers at EMBL’s European Bioinformatics Institute (EMBL-EBI), Cold Spring Harbor Laboratory (CSHL), and the Massachusetts Institute of Technology (MIT), the research delves into why some tumors remain unresponsive to immune checkpoint blockade (ICB) therapy, an approved treatment that utilizes the patient’s immune system to attack and eliminate cancer cells.
ICB has transformed the treatment landscape for cancer patients. Response rates range between 15% and 60%, but it is still unclear why some patients do not respond. Understanding what happens at the cellular level could help clinicians predict which patients are more likely to respond and guide treatment decisions. ICB is known to be most effective in DNA mismatch repair deficient (MMRd) tumors, but still only half of the MMRd tumors respond to ICB, and among responders, many will unfortunately relapse. This study looks at the complex mechanisms underlying response to ICB in patients with MMRd tumors.
ICB functions by obstructing an immune checkpoint – a signal exploited by cancer cells to stop the immune system from detecting the tumor through the high number of mutations found within these cancer cells. Such mutations can serve as cues that enable the immune system to identify and combat the tumor. In the context of ICB, weaker mutation signals lead to a diminished response to treatment because the immune system has a harder time finding and recognizing the cancer cells.
The findings of this study, published in the journal Nature Genetics, highlight the pivotal role played in this process by intratumoral heterogeneity.
“This is an important body of work that provides new insights into the factors that control immune responses against cancer and why some tumors fail to respond to immune-stimulating therapies,” said Tyler Jacks, Professor at the Koch Institute at MIT.
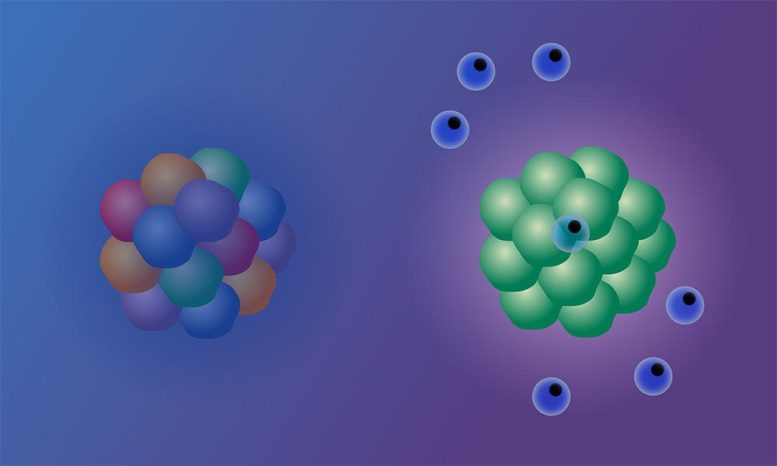
“One way to picture this is to imagine a crowd, where each person is holding a yellow flashlight,” explained Isidro Cortes-Ciriano, Research Group Leader at EMBL-EBI. “If everyone turns on their flashlight, the beam of yellow light can be seen from far away. Similarly, the more cells with the same mutations in a tumor, the stronger the signal and the more likely to trigger an immune response. However, if each person in the crowd has a different colored flashlight, the light emanating from the crowd is less clear, and the signal becomes jumbled. Similarly, if cancer cells have different mutations, the signal is harder to make out and the immune system is not triggered, so ICB doesn’t work.”
Understanding Immunotherapy Response
ICB has shown remarkable efficacy in tumors with a high number of mutations, In particular, this applies to tumors with clonal neoantigens. Clonal neoantigens occur when identical mutations are present across all cells of a tumor. Despite this, less than half of MMRd tumors show long-lasting responses to ICB, posing a significant challenge in optimizing treatment.
This study dissects the molecular mechanisms causing resistance to ICB in MMRd tumors and shows that intratumoral heterogeneity – a wide variety of mutations spread across the tumor – dampens the immune response, leading to diminished effectiveness of the ICB treatment.
“Our goal was to unravel the mystery of why certain tumors, which should respond to immunotherapy, do not,” said Peter Westcott, Assistant Professor at Cold Spring Harbor Laboratory, former Postdoctoral Researcher at MIT. Regarding the tumors in their study, Westcott said, “There’s no question these tumors are MMRd, yet they’re not responding. That is a profoundly interesting negative result. By studying the mechanisms behind this resistance, we can pave the way for the development of more effective and personalized treatment strategies.”
Improving Clinical Practices
The findings of this study provide a means to identify which patients are more likely to benefit from ICB treatment, highlighting the need for personalized treatment approaches. In their investigation, the researchers used mouse models to demonstrate that inactivation of MMR is not enough to improve patient responsiveness to ICB.
“Our understanding of cancer is improving all the time, and this translates into better patient outcomes,” added Cortes-Ciriano. “Survival rates following a cancer diagnosis have significantly improved in the past twenty years, thanks to advanced research and clinical studies. We know that each patient’s cancer is different and will require a tailored approach. Personalized medicine must take into account new research that is helping us understand why cancer treatments work for some patients but not all.”
Access to Clinical Data
The study used preclinical models, including mouse models and cell lines, as well as clinical trial data from colon and gastric cancer patients, to study and analyze tumor responses to ICB.
Using clinical data, the researchers observed that colon and stomach tumors with a diluted mutational signal caused by intratumoral heterogeneity displayed reduced sensitivity to ICB treatment. This finding also suggests that identifying the level of signal strength in individual tumors could help predict a patient’s response to ICB in the clinic.
“One of the major challenges of the study was getting access to clinical trial data,” explained Isidro Cortes-Ciriano. “This highlights once again how important it is for research data to be accessible via secure mechanisms so it can be reused to uncover new insights and improve our understanding of disease.”
Reference: “Mismatch repair deficiency is not sufficient to elicit tumor immunogenicity” by Peter M. K. Westcott, Francesc Muyas, Haley Hauck, Olivia C. Smith, Nathan J. Sacks, Zackery A. Ely, Alex M. Jaeger, William M. Rideout III, Daniel Zhang, Arjun Bhutkar, Mary C. Beytagh, David A. Canner, Grissel C. Jaramillo, Roderick T. Bronson, Santiago Naranjo, Abbey Jin, J. J. Patten, Amanda M. Cruz, Sean-Luc Shanahan, Isidro Cortes-Ciriano and Tyler Jacks, 14 September 2023, Nature Genetics.
DOI: 10.1038/s41588-023-01499-4
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.