New research reveals how skin-derived signals may help turn local stress into a body-wide antibody response.
Skin does much more than act as a protective covering. New research tackles a basic question in immunology: how can a skin infection or injury in one spot trigger a strong body-wide antibody response that helps stop pathogens from spreading?
The researchers, including collaborators from the Chinese Academy of Medical Sciences Institute of Dermatology and Tsinghua’s School of Pharmaceutical Sciences, point to keratinocytes as a key part of the answer.
The research teams, including collaborators from the Chinese Academy of Medical Sciences Institute of Dermatology and Tsinghua’s School of Pharmaceutical Sciences, point to keratinocytes as a key part of the answer.
In the first study, published in Nature, the researchers found that keratinocytes quickly build up a metabolic intermediate called farnesyl pyrophosphate (FPP) from the mevalonate pathway after skin infection or ultraviolet exposure. This increase is driven by the unfolded protein response, which activates SREBF transcription factors.
FPP then binds directly to the intracellular domain of TRPV3 in keratinocytes, causing calcium influx. That calcium signal activates two downstream pathways, Ca2+–CaM–calcineurin–NFAT and PYK2–RAS–ERK, which then stimulate production of the immune mediators IL-6 and CCL20.

From Local Stress to Systemic Antibody Production
IL-6 supports the development of T follicular helper cells, while CCL20 attracts migratory dendritic cells to the draining lymph nodes. Together, these signals strongly boost germinal center reactions, leading to robust systemic production of pathogen-specific IgG antibodies and the formation of long-lived memory B cells and plasma cells. In this way, FPP acts as an endogenous “alarmin” that sends out a local danger signal and helps activate the broader adaptive immune system.
A companion study, published in Immunity & Inflammation, adds an important pharmacological angle. The team found that two aromatic compounds from medicinal plants, Carvacrol and Camphor, which are known for their characteristic smells, can function as titratable organic adjuvants. When given locally with an antigen, the compounds significantly increased antigen-specific IgG responses in a dose-dependent manner, with no observed toxicity at effective doses in mouse models.
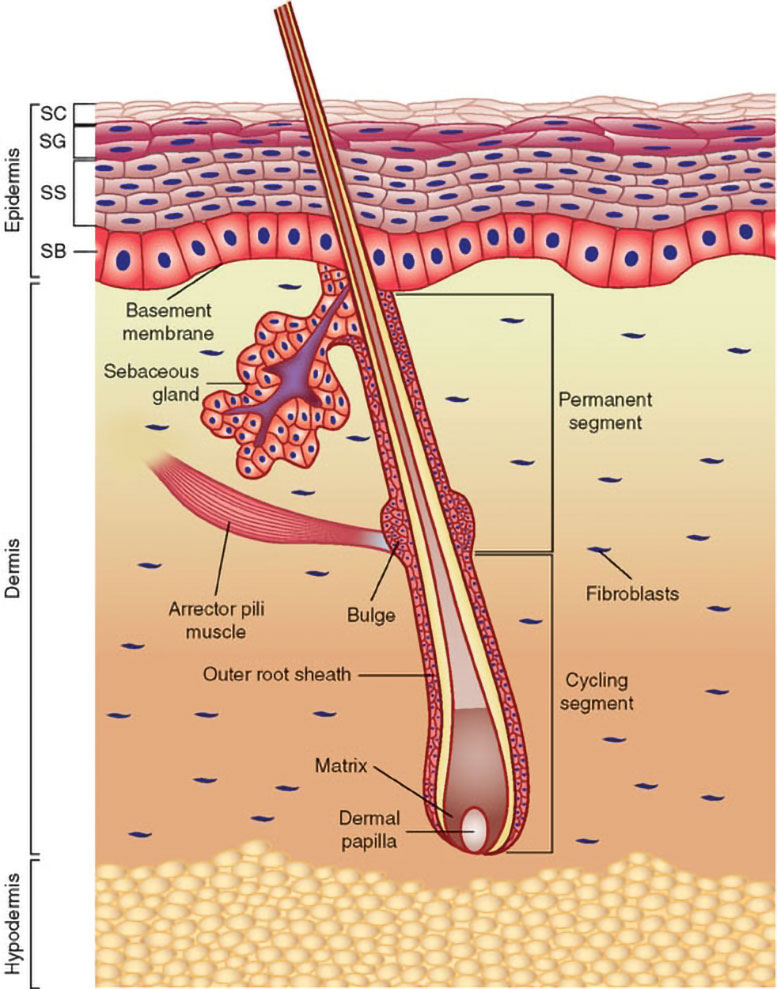
A Distinct Route to the Same Immune Pathway
Carvacrol and Camphor also activate TRPV3 in keratinocytes, leading to calcium influx and increased expression of IL-6, CCL20, and TNF. Importantly, the endogenous activator FPP must bind to specific intracellular sites on TRPV3 (residues R416 and K581), while these external fragrant agonists activate the channel through a separate mechanism. This two-part activation model, one metabolic and one sensory, underscores TRPV3’s role as a molecular hub that can integrate different kinds of signals.
“These findings reveal an elegant system where internal metabolic alarms and external sensory cues converge on the same pathway to calibrate immune responses,” explained Professor Liu. “The fact that simple, plant-derived fragrant molecules can quantitatively boost antibody production opens up exciting new avenues for vaccine adjuvant design.”
Therapeutic and Vaccine Implications
The findings could have broad implications. First, they identify TRPV3, along with the downstream effectors IL-6 and CCL20, as potential therapeutic targets for autoimmune diseases such as systemic lupus erythematosus, where abnormal B cell responses contribute to disease.
Second, they introduce a new class of adjuvants: titratable organic compounds that work locally to strengthen systemic humoral immunity. Unlike traditional adjuvants, which often cause broad inflammation, these fragrant TRPV3 agonists may offer a more targeted and potentially safer way to improve vaccine performance, especially for mucosal and anti-infective vaccines.
By linking metabolism, skin biology, and humoral immunity, the studies provide a foundation for better understanding immune crosstalk and for developing next-generation immunotherapies.
References:
“A metabolic alarmin from keratinocytes potentiates systemic humoral immunity” by Zhenglin Ji, Ji Gao, Shaocun Zhang, Jiajie Li, Haijing Wu, Jing Yao, Xianqiang Ma, Yue Xin, Yongjie Zhu, Meng Zhao, Zhidan Zhao, Kai Shen, Tao Wu, Xinmin Qian, Juanjuan Wang, Haoran An, Yuxin Li, Wenbo Sun, Qiancheng Zhao, Xiaoying Zhou, Ruiyu Gao, Qinghui Duan, Cuifeng Li, Xiaoke Geng, Ming Yang, Rong Xiao, Juan Liu, Wang Wang, Ji Wang, Yesheng Fu, Jing-Ren Zhang, Xiangjun Chen, Pei Tong, Gong Cheng, Hai Qi, Li Wu, Wenwen Zeng, Qiaoran Xi, Lingqiang Zhang, Yuping Lai, Wei Yang, Yonghui Zhang, Qianjin Lu and Wanli Liu, 4 March 2026, Nature.
DOI: 10.1038/s41586-026-10167-6
“Fragrant TRPV3 agonists act as titratable organic adjuvants to amplify antigen-specific IgG response” by Zhenglin Ji, Ji Gao, Yue Xin, Cuifeng Li, Qianjin Lu, Haijing Wu and Wanli Liu, 5 March 2026, Immunity & Inflammation.
DOI: 10.1007/s44466-026-00033-5
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.