A new method for creating one of chemistry’s most widely used class of compounds could revolutionize industrial processes, making them cheaper, simpler, and more environmentally friendly.
A group of researchers led by scientists at Hokkaido University have developed a simpler, greener method for producing Grignard reagents—one of the most important and widely used type of reagents in the chemical industry—that drastically cuts down on the use of hazardous organic solvents and could lead to reduced production costs. This new process was reported in Nature Communications.

Grignard reagents are an essential ingredient in a common method for creating carbon-carbon bonds, the building blocks of organic molecules. These reagents were discovered 120 years ago, but due to their instability, the conventional production method still used today is carried out in toxic organic solvents with no exposure to moisture and oxygen. This results in a complicated, delicate, and expensive process that produces environmentally hazardous waste.
Researchers sidestepped these problems by minimizing the amount of organic solvent used and by employing a mechanochemical technique called ball-milling to produce Grignard reagents. The reactants, magnesium metal and organohalides, were loaded into a metal chamber along with a stainless-steel ball. In a key step, a small amount of organic solvent—about one-tenth the amount used in conventional methods—was added to the solid reactants. The chamber was then spun for one hour, causing the ball to tumble around and slam into the solid-state reactants, helping them to mix thoroughly and react, forming a paste-like Grignard reagent.
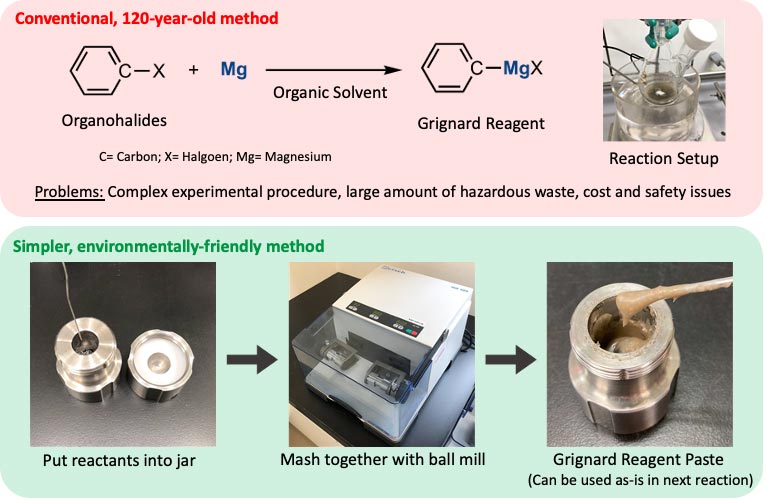
Researchers even succeeded in creating new Grignard reagents using organohalides that have poor solubility in organic solvents, which can’t typically be made by the conventional method. Avoiding heavy use of organic solvents allowed the researchers to overcome solubility problems, which opens up a world of new reactions with Grignard reagents prepared from insoluble compounds. It also leads to a major reduction in hazardous waste.
Additionally, it is more difficult for water or oxygen to affect the Grignard reagents when less organic solvent is used. This means that removing water and oxygen from the surrounding air is not required, making the process easier to perform and less costly. Given the potential economic and environmental benefits, this discovery could have a huge effect on chemical industries.

“With a growing need to address environmental concerns and reduce CO2 emissions, it is important to develop chemical reactions that don’t require organic solvents,” commented Associate Professor Koji Kubota. “Grignard reagents are arguably the most well-known, commonly used reagents in industry, and so our work could fundamentally change the way a vast number of chemicals are produced at scale, leading to significantly reduced impact on the environment.”
Reference: “Mechanochemical synthesis of magnesium-based carbon nucleophiles in air and their use in organic synthesis” by Rina Takahashi, Anqi Hu, Pan Gao, Yunpeng Gao, Yadong Pang, Tamae Seo, Julong Jiang, Satoshi Maeda, Hikaru Takaya, Koji Kubota and Hajime Ito, 18 November 2021, Nature Communications.
DOI: 10.1038/s41467-021-26962-w
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.