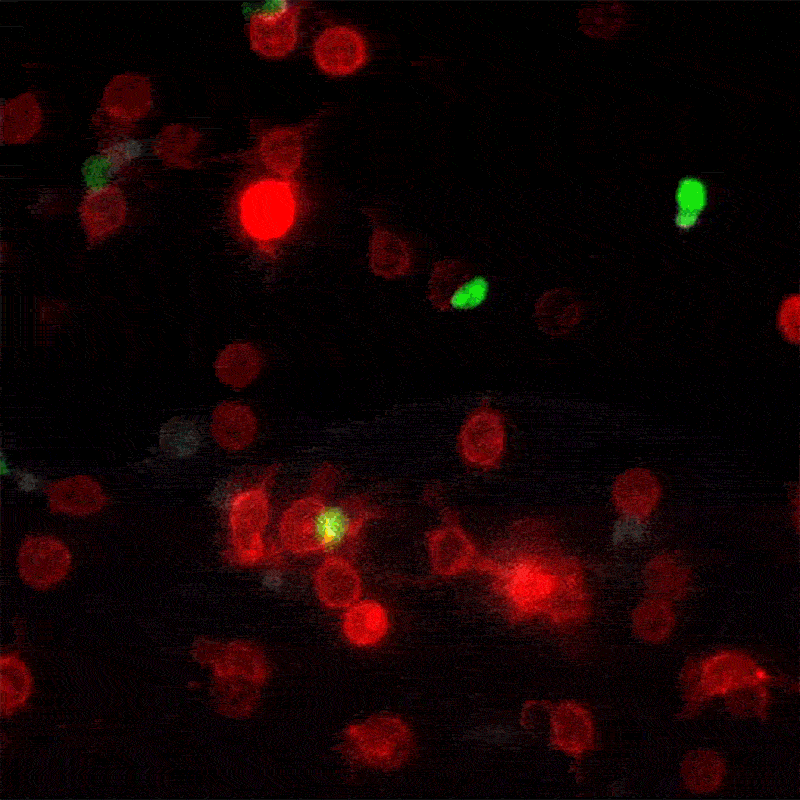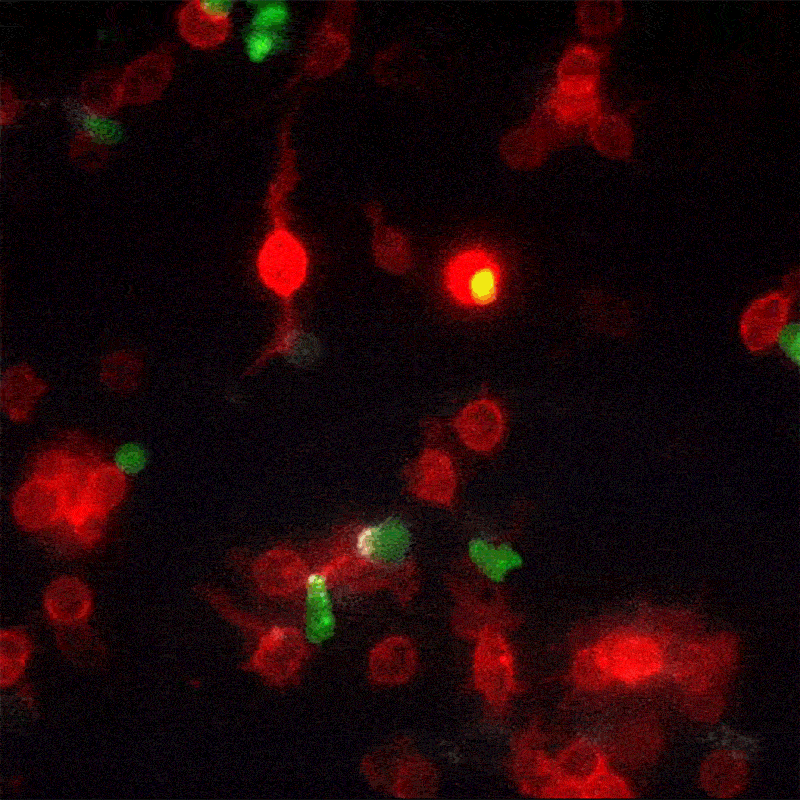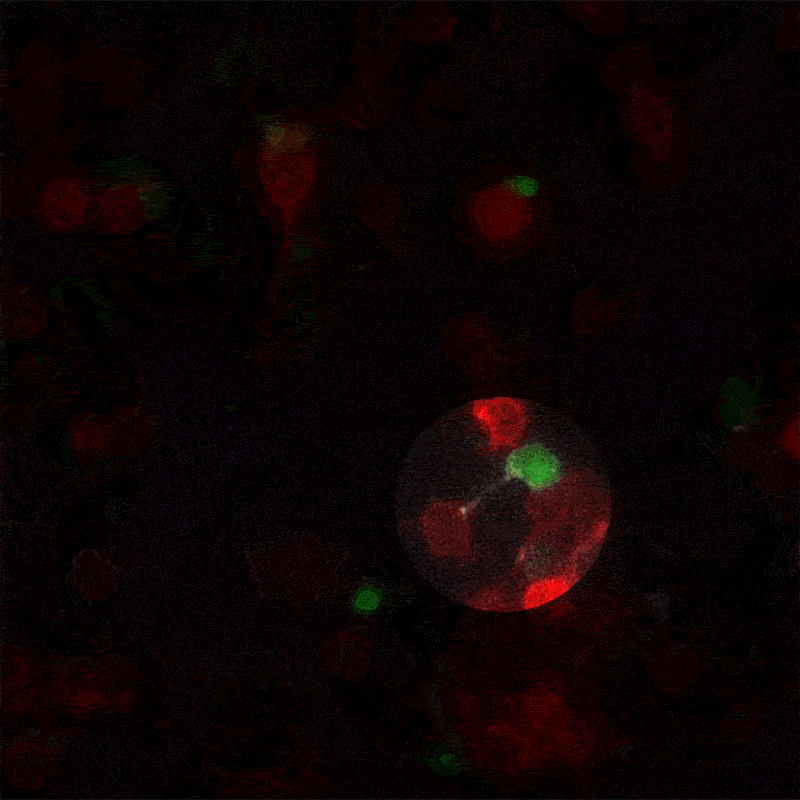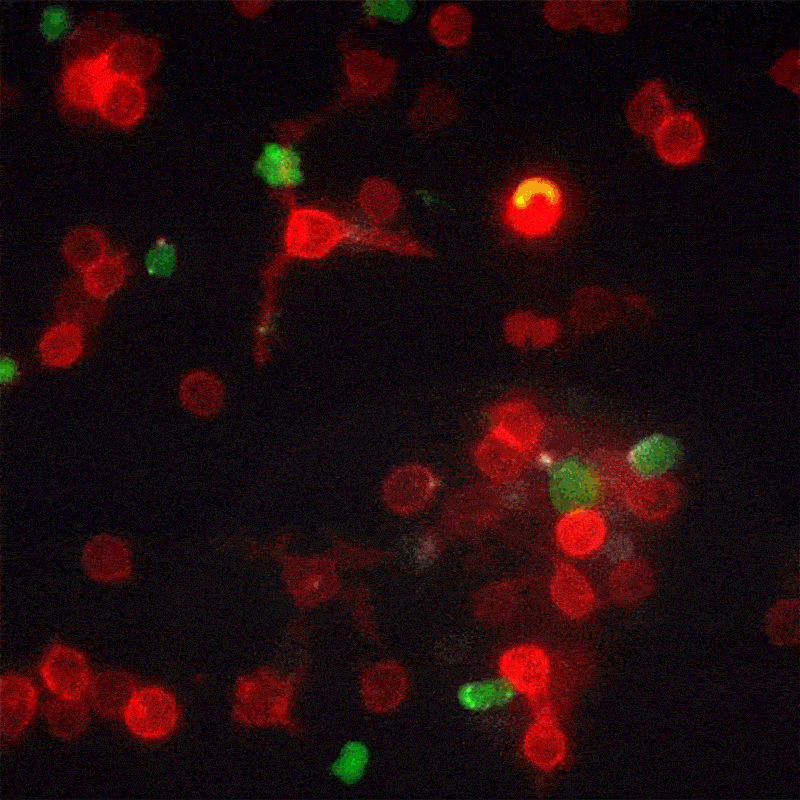
A new method for monitoring interactions between cells, dubbed LIPSTIC by its creators, is much more than a cosmetic improvement over existing techniques. The breakthrough, led by The Rockefeller University assistant professor Gabriel Victora, offers scientists in a wide range of disciplines a powerful new tool for studying complex biological events as they play out in live animals.
“Now we have a way to tell whether one type of cell has been communicating with another cell within a live mouse,” says Victora, an immunologist and head of the Laboratory of Lymphocyte Dynamics. Until now, scientists were able to study such exchanges between cells only within the artificial environment of a petri dish.
“Virtually all of immunology is based on cell-to-cell contact,” says Victora. “Most immune phenomena require that cells physically meet each other and exchange signals so that the response gets kick started. That is how the system decides whether or not it wants to react to a stimulus like an invasion by a pathogen, and if it is going to act, what exactly it should do.”
Telltale stains
In his most recent paper, published this month in the journal Nature, Victora refers to these brief encounters between immune cells as “kiss-and-run:” within a crowd of cells that all look the same, individual ones will occasionally engage in talking to another cell, then move away. To track these temporary interactions, Victora and his team, including postdoctoral associate and first author Giulia Pasqual, devised a system for labeling the cellular structures that actually make physical contact when two cells meet.
When two such manipulated cells interact, they tag each other with the biological equivalent of a smear of lipstick. Thus marked, the cells actively involved in an immune response can be readily identified and counted with flow cytometry, a standard lab technology.
“This enables us to figure out which exact cells within a population are interacting,” says Victora. “We can also monitor how the number and quality of the interacting cells changes over time.” This trove of information, unavailable until now, can help researchers better understand how the immune system functions in the presence of a foreign body.
To develop and test the new method, the researchers worked with two kinds of immune-system cells: T-cells, the “executive directors” of the immune response, and dendritic cells, which alert the T-cells to the presence of pathogens by picking up fragments of the invaders known as antigens and presenting them to the T-cells through direct cell-to-cell contact.
A test drive
Once they confirmed that the labeling system worked in the lab, they tested it in mice. For those experiments, Victora picked a classic model of the immune response that involves loading dendritic cells with an antigen that the T-cells recognize via receptors on their surfaces, then injecting these primed dendritic cells into a mouse’s foot. From there, the dendritic cells travel to the nearest lymph node, where T-cells reside.
When the researchers processed and analyzed the lymph nodes from these mice using flow cytometry, they found that the labeled immune cells in the live mice had indeed tagged each other while interacting in the lymph node, and that those cells could be identified and counted.
“Now we can measure things that we couldn’t measure before,” Victora says, describing, in his succinct, understated way, exactly the sort of landmark achievement that drives all science.
Reference: “Monitoring T cell–dendritic cell interactions in vivo by intercellular enzymatic labelling” by Giulia Pasqual, Aleksey Chudnovskiy, Jeroen M. J. Tas, Marianna Agudelo, Lawrence D. Schweitzer, Ang Cui, Nir Hacohen and Gabriel D. Victora, 17 January 2018, Nature.
DOI: 10.1038/nature25442
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.