Llama antibodies clamp the coronavirus shut at its weakest spot—leaving the virus powerless to mutate or infect.
Researchers have identified a special type of small antibody that offers strong protection against a wide variety of SARS-related coronaviruses. These include the original SARS-CoV-1 as well as multiple versions of SARS-CoV-2, the virus that causes COVID-19. The antibodies work by attaching to a highly conserved and essential region located at the base of the virus’s spike protein. This action prevents the spike from changing shape, which is necessary for the virus to infect cells. The study, published in Nature Communications, could help pave the way for broad antiviral treatments that stay effective even as new variants emerge.
Why New Variants Make Antibody Treatments Less Effective
As SARS-CoV-2 continues to evolve, it produces new variants that can evade many existing antibody therapies. This is because most antibodies are designed to target regions of the spike protein that frequently mutate, like the receptor binding domain. When these regions change, antibodies may no longer recognize the virus, making treatments less effective or even useless.
To get around this problem, scientists led by Professor Xavier Saelens and Dr. Bert Schepens at the VIB-UGent Center for Medical Biotechnology decided to focus on a different part of the spike protein. They targeted the S2 subunit, which plays a key role in helping the virus fuse with human cells. Unlike other parts of the spike protein, the S2 subunit is much more stable across different types of coronaviruses.
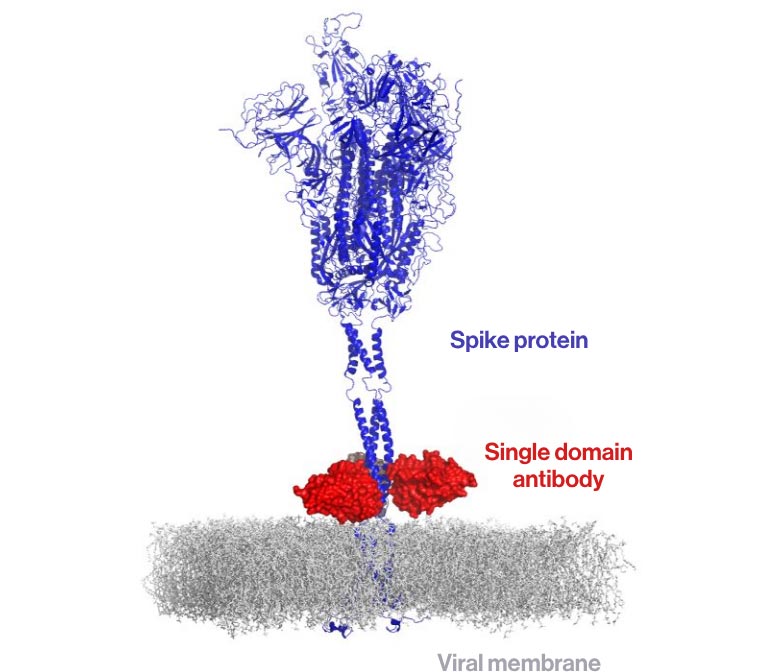
Llama Antibodies Provide a Powerful New Tool
To find the right kind of antibodies, the team turned to llamas—specifically, one named Winter. Llamas produce single-domain antibodies (also known as VHHs or nanobodies) that are much smaller than typical human antibodies. The researchers discovered several of these llama-derived nanobodies that could neutralize a broad range of SARS-related viruses.
What sets these antibodies apart is how they work. Instead of targeting the virus’s usual weak spots, they act like a clamp. They attach to a rarely exposed, tightly conserved region made up of three twisted protein structures (called a coiled coil of alpha helices) at the base of the spike protein. By binding to this region, the antibodies lock the spike in its original shape and block it from unfolding—an essential step in the infection process.
Strong Protection, Even When the Virus Tries to Fight Back
In lab tests, these antibodies provided strong protection against infection in animals, even when given in small amounts. When researchers tried to force the virus to evolve and escape the treatment, it had a hard time doing so. The few variants that did emerge were much less infectious, suggesting that the virus cannot easily mutate this region without losing its ability to spread effectively.
“This region is so crucial to the virus that it can’t easily mutate without weakening the virus itself,” said Dr. Schepens, the study’s senior author. “That gives us a rare advantage: a target that’s both essential and stable across variants.”

Hope for Long-Lasting COVID and Coronavirus Treatments
This discovery marks an important step toward developing long-lasting antiviral therapies that could remain effective against current and future coronavirus threats.
“The combination of high potency, broad activity against numerous viral variants, and a high barrier to resistance is incredibly promising,” said Prof. Saelens. “This work provides a strong foundation for developing next-generation antibodies that could be vital in combating not only current but also future coronavirus threats.”
Reference: “Ultrapotent SARS coronavirus-neutralizing single-domain antibodies that clamp the spike at its base” by Sieglinde De Cae, Inge Van Molle, Loes van Schie, Sophie R. Shoemaker, Julie Deckers, Nincy Debeuf, Sahine Lameire, Wim Nerinckx, Kenny Roose, Daria Fijalkowska, Simon Devos, Anne-Sophie De Smet, Jackeline Cecilia Zavala Marchan, Toon Venneman, Koen Sedeyn, Lejla Mujanovic, Marlies Ballegeer, Manon Vanheerswynghels, Caroline De Wolf, Hans Demol, Jasper Zuallaert, Pieter Vanhaverbeke, Gholamreza Hassanzadeh Ghassabeh, Chiara Lonigro, Viki Bockstal, Manuela Rinaldi, Rana Abdelnabi, Johan Neyts, Susan Marqusee, Bart N. Lambrecht, Nico Callewaert, Han Remaut, Xavier Saelens and Bert Schepens, 30 May 2025, Nature Communications.
DOI: 10.1038/s41467-025-60250-1
Funding: Fonds Wetenschappelijk Onderzoek, EOS-programme, EU Horizon 2021, Exevir Bio BV
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
This is a wonderfully important discovery by these dedicated researchers.
They aught to be universally commended for their work.
However , there were at least two other very similar discoveries that briefly made headline news in online medical science circles way back in the early months of the Covid-19 pandemic in 2020 and 2021 which held similar promise for mitigating illness and death .
The first was a lama- based nano body inhaler dubbed “AeroNabs.”
Developed by UCSF researchers , it was reportedly capable of preventing infection by blocking spike adhesion in the nasal passages.
https://www.ucsf.edu/news/2020/08/418241/aeronabs-promise-powerful-inhalable-protection-against-covid-19
Also a lama-derived nanobody medicine discovered by University of Pittsburgh researchers that looked as if it could have become an important preventive treatment in the early period of this horrible and deadly scourge on the world.
https://www.upmc.com/media/news/110520-shi-llama-nanobody
Neither saw the popular light of day despite their very promising development and testing results.