
Prostaglandin class drugs are a class of hormone-like chemicals that influence processes including blood circulation and inflammation. Their use offers a way to churn out different versions of the molecules thanks to organocatalysis, an organic chemistry discipline that speeds up chemical reactions with the help of carbon-based molecules to avoid the drawbacks of metal catalysts.
The findings, published in the journal Nature, provide with researchers an easier way of accessing the family of molecules that includes prostaglandin. Previously, it took as much as 20 chemical steps to synthesize these types of drugs. The new synthesis described could build a prostaglandin molecule in as little as seven steps. Synthetic chemist Varinder Aggarwal, of the University of Bristol in the UK, realized that he could simplify the process by using the amino acid (S)-proline as an organocatalyst to make sure that the end result had the exact same shape.
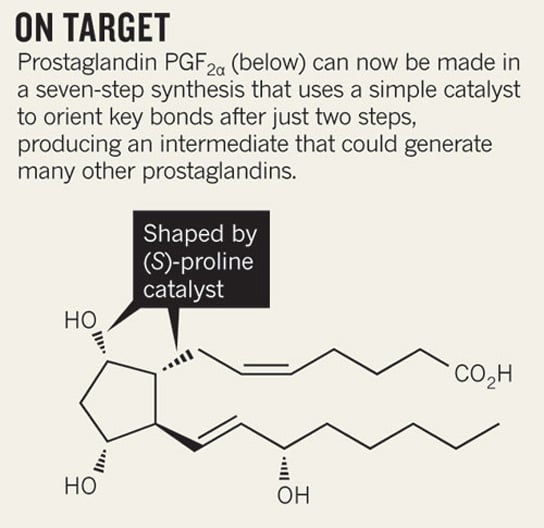
The synthesized molecule in Aggarwal’s reaction could be used as a foundation to build the whole family of prostaglandin drugs. The reaction was even to create PGF2α, the most complicated prostaglandin. It’s also specific enough to produce left- or right-handed versions of the molecule, which is crucial in making an effective drug.
The reaction is viable commercially since it doesn’t use any expensive and toxic metals as catalysts. However, further development is needed before it can be used to produce drugs on vaster scales.
Reference: “Stereocontrolled organocatalytic synthesis of prostaglandin PGF2α in seven steps” by Graeme Coulthard, William Erb and Varinder K. Aggarwal, 15 August 2012, Nature.
DOI: 10.1038/nature11411
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.