A study discovered that Alzheimer’s disease symptoms are linked to the depletion of the normal soluble protein rather than its aggregation into plaques.
Scientists at University of Cincinnati say restoring a brain protein, not removing amyloid plaques, should be the target of Alzheimer’s dementia therapies.
Experts estimate more than 6 million Americans are living with Alzheimer’s dementia. But a recent study, led by the University of Cincinnati, sheds new light on the disease and a highly debated new drug therapy.
The UC-led study, conducted in collaboration with the Karolinska Institute in Sweden, claims that the treatment of Alzheimer’s disease might lie in normalizing the levels of a specific brain protein called amyloid-beta peptide. This protein is needed in its original, soluble form to keep the brain healthy, but sometimes it hardens into “brain stones” or clumps, called amyloid plaques.
The study, which appears in the journal EClinicalMedicine (published by the Lancet), comes on the heels of the FDA’s conditional approval of a new medicine, aducanumab, that treats the amyloid plaques.
“It’s not the plaques that are causing impaired cognition,” says Alberto Espay, the new study’s senior author and professor of neurology at UC. “Amyloid plaques are a consequence, not a cause,” of Alzheimer’s disease, says Espay, who is also a member of the UC Gardner Neuroscience Institute.
Alzheimer’s disease became widely known as “the long goodbye” in the late 20th century due to the disease’s slow deterioration of brain function and memory. It was over 100 years ago, however, that scientist Alois Alzheimer first identified plaques in the brain of patients suffering from the disease.
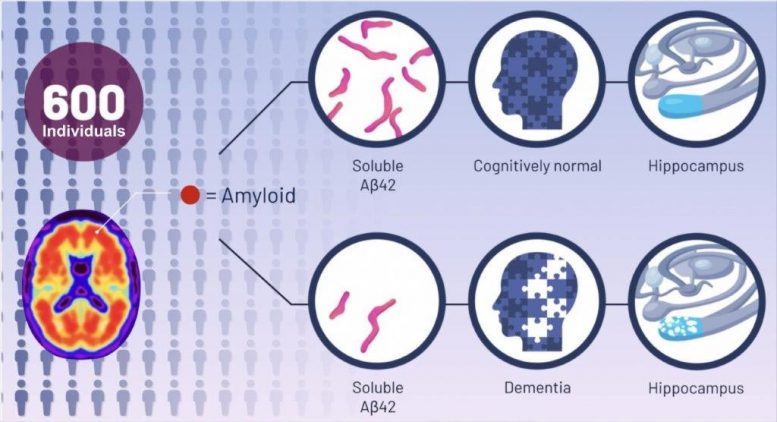
Since then, Espay says that scientists have focused on treatments to eliminate the plaques. But the UC team, he says, saw it differently: Cognitive impairment could be due to a decline in soluble amyloid-beta peptide instead of the corresponding accumulation of amyloid plaques. To test their hypothesis, they analyzed the brain scans and spinal fluid from 600 individuals enrolled in the Alzheimer’s Disease Neuroimaging Initiative study, who all had amyloid plaques. From there, they compared the amount of plaques and levels of the peptide in the individuals with normal cognition to those with cognitive impairment. They found that, regardless of the amount of plaques in the brain, the individuals with high levels of the peptide were cognitively normal.
They also found that higher levels of soluble amyloid-beta peptide were associated with a larger hippocampus, the area of the brain most important for memory.
According to the authors, as we age most people develop amyloid plagues, but few people develop dementia. In fact, by the age of 85, 60% of people will have these plagues, but only 10% develop dementia, they say.

Alberto Espay, MD, MSc, professor of neurology at the UC College of Medicine and Director and Endowed Chair of the James J. and Joan A. Gardner Family Center for Parkinson’s Disease and Movement Disorders. Credit: Colleen Kelley/UC Brand + Creative
“The key discovery from our analysis is that Alzheimer’s disease symptoms seem dependent on the depletion of the normal protein, which is in a soluble state, instead of when it aggregates into plaques,” says co-author Kariem Ezzat from the Karolinska Institute.
The most relevant future therapeutic approach for the Alzheimer’s program will be replenishing these brain soluble proteins to their normal levels, says Espay.
The research team is now working to test their findings in animal models. If successful, future treatments may be very different from those tried over the last two decades. Treatment, says Espay, may consist of increasing the soluble version of the protein in a manner that keeps the brain healthy while preventing the protein from hardening into plaques.
Reference: “High cerebrospinal amyloid-β 42 is associated with normal cognition in individuals with brain amyloidosis” by Andrea Sturchio, Alok K. Dwivedi, Christina B. Young, Tarja Malm, Luca Marsili, Jennifer S. Sharma, Abhimanyu Mahajan, Emily J. Hill, Samir EL Andaloussi, Kathleen L. Poston, Fredric P. Manfredsson, Lon S. Schneider, Kariem Ezzat and Alberto J. Espay, 28 June 2021, EClinicalMedicine.
DOI: 10.1016/j.eclinm.2021.100988
Co-authors include: Andrea Sturchio, University of Cincinnati, and Samir EL Andaloussi, Karolinska Institute.
The research was funded by the UC Gardner Neuroscience Institute.
The authors disclose that they have recently cofounded REGAIN Therapeutics, owner of a patent application that covers synthetic soluble non-aggregating peptide analogues as replacement treatment in proteinopathies.







This is very good news. I hope it becomes an available treatment soon, since I am 72 and worry about cognitive decline in my future. Not many in my family have had dementia and my paternal grandmother alived to the age of 99 with no dementia, but I don’t believe in pushing my luck. Congratulations on the discovery.
Alzheimer’s disease treated with success is a good achivement in medical science.While synthesizing the quite artificial brain protein solution in the laboratory neuro-stae &function of a patient can be checked with some regular time intervals,so as to confirm improvement in health condition.So other harmful diseases affecting vital organs of the body along with various mental disrass have large impact.
As a health care professional this could be game changing paradigm shift. I cannot overstate the importance of this finding from the UC researchers. Wow…..
Ok. How do we restore the Amyloid-Beta Peptides?
Can progress become front page news with each study and recommendation. So useful!!
This sounds like a great achievement. As I have Alzeimers in my family it is very encouraging at the same time I wonder if this will have any benefit for childhood dementia.
Check your spelling. A few times you mean to say “plaque”, but write “plague”.