High-risk heart patients show improved outcomes and reduced bleeding by over 50% when aspirin is withdrawn one month after PCI, with ticagrelor treatment alone, according to the ULTIMATE-DAPT study.
Withdrawing aspirin one month after percutaneous coronary intervention (PCI) in high-risk heart patients and keeping them on ticagrelor alone safely improves outcomes and reduces major bleeding by more than half when compared to patients taking aspirin and ticagrelor combined (also known as dual antiplatelet therapy or DAPT), which is the current standard of care.
These are the results from the ULTIMATE-DAPT study announced during a late-breaking trial presentation at the American College of Cardiology Scientific Sessions on April 7, and published in The Lancet.
Study Findings and Implications
This is the first and only trial to test high-risk patients with recent or threatened heart attack (acute coronary artery syndromes, or ACS) taking ticagrelor with a placebo starting one month after PCI, and compare them with ACS patients taking ticagrelor with aspirin over the same period. The significant findings could change the current guidelines for standard of care worldwide.
“Our study has demonstrated that withdrawing aspirin in patients with recent ACS one month after PCI is beneficial by reducing major and minor bleeding through one year by more than 50 percent. Moreover, there was no increase in adverse ischemic events, meaning continuing aspirin was causing harm without providing any benefit,” says Gregg W. Stone, MD, the study co-chair of ULTIMATE-DAPT, who presented the trial results.
“It is my belief that it’s time to change the guidelines and standard clinical practice such that we no longer treat most ACS patients with dual antiplatelet therapy beyond one month after a successful PCI procedure. Treating these high-risk patients with a single potent platelet inhibitor such as ticagrelor will improve prognosis,” adds Dr. Stone, who is Director of Academic Affairs for the Mount Sinai Health System and Professor of Medicine (Cardiology), and Population Health Science and Policy, at the Icahn School of Medicine at Mount Sinai.
Study Details and Results
The study analyzed 3,400 patients with ACS at 58 centers in four countries between August 2019 and October 2022. All of the patients had undergone PCI, a non-surgical procedure in which interventional cardiologists use a catheter to place stents in the blocked coronary arteries to restore blood flow. The patients were stable one month after PCI and were on ticagrelor and aspirin. Researchers randomized the patients after one month, withdrawing aspirin in 1,700 patients and putting them on ticagrelor and a placebo, while leaving the other 1,700 patients on ticagrelor and aspirin. All patients were evaluated between 1 and 12 months after the procedure.
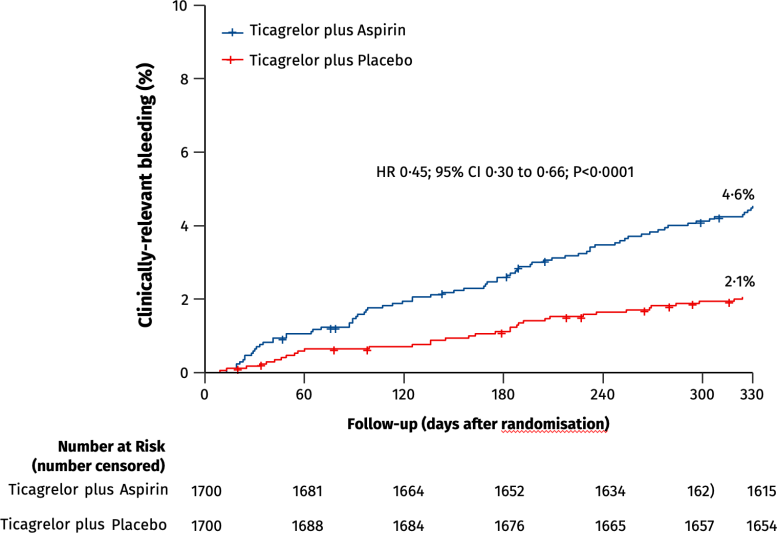
During the study period, 35 patients in the ticagrelor-placebo group had a major or minor bleeding event, compared to 78 patients in the ticagrelor-aspirin group, meaning that the incidence of overall bleeding incidents was reduced by 55 percent by withdrawing aspirin. The study also analyzed major adverse cardiac and cerebrovascular events including death, heart attack, stroke, bypass graft surgery, or repeat PCI. These events occurred in 61 patients in the ticagrelor-placebo group compared to 63 patients in the ticagrelor-aspirin group, and were not statistically significant – further demonstrating that removing aspirin did no harm and improved outcomes.
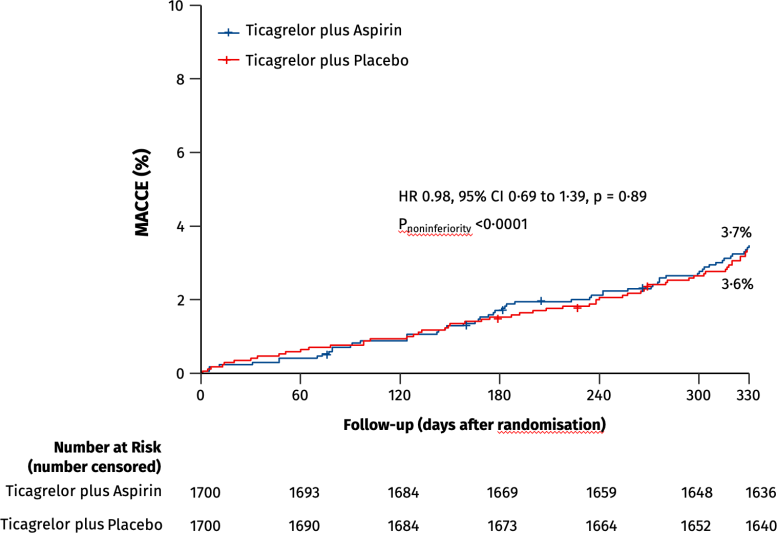
PCI denotes percutaneous coronary intervention; BARC denotes Bleeding Academic Research Consortium; MACCE, major adverse cardiovascular or cerebrovascular events; HR, hazard ratio; CI, confidence interval.
Credit: Mount Sinai Health System
“It was previously believed that discontinuing dual antiplatelet therapy within one year after PCI in patients with ACS would increase the risk of heart attack and other ischemic complications, but the present study shows that is not the case, with contemporary drug-eluting stents now used in all PCI procedures. Discontinuing aspirin in patients with a recent or threatened heart attack who are stable one month after PCI is safe and, by decreasing serious bleeding, improves outcomes,” Dr. Stone adds. “This study extends the results of prior work that showed similar results but without the quality of using a placebo, which eliminates bias from the study.”
Reference: “Ticagrelor alone versus ticagrelor plus aspirin from month 1 to month 12 after percutaneous coronary intervention in patients with acute coronary syndromes (ULTIMATE-DAPT): a randomised, placebo-controlled, double-blind clinical trial” by Zhen Ge, Jing Kan, Xiaofei Gao, Afsar Raza, Jun-Jie Zhang, Bilal S Mohydin, Fentang Gao, Yibing Shao, Yan Wang, Hesong Zeng, Feng Li, Hamid Sharif Khan, Naeem Mengal, Hongliang Cong, Mingliang Wang, Lianglong Chen, Yongyue Wei, Feng Chen, Gregg W Stone, Shao-Liang Chen, Xiaobo Li, Zhen Ge, Jing Kan, Muhammed Anjum, Fei Ye, Xiaofei Gao, Anjum Jalal, Ping Xie, Ling Tao, Xiang Chen, Hamid S Khan, Asim Javed, Yibin Shao, Xiaomei Guo, Feng Li, Tahir Saghir, Naeem Mengal, Shaoping Nie, Hong Qu, Xuesong Qian, Song Yang, Jing Chen, Dasheng Gao, Lijun Liu, Mingliang Wang, Lianglong Chen, Fan Liu, Tan Xu, Yinwu Liu, Badar Ul Ahad Gill, Qing Yang, Nin Guo, Shangyu Wen, Hongliang Cong, Lang Hong, Imad Sheiban, Afsar Raza, Yongyue Wei, Feng Chen, Gary S Mintz, Jun-Jie Zhang, Gregg W Stone and Shao-Liang Chen, 7 April 2024, The Lancet.
DOI: 10.1016/S0140-6736(24)00473-2
This trial was funded by the Chinese Society of Cardiology, the National Natural Scientific Foundation of China, and Jiangsu Provincial & Nanjing Municipal Clinical Trial Project.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.