A new study indicates that male DNA, left over from pregnancy with a male fetus, can persist in a woman’s brain throughout the rest of her life. Although the exact biological impact of the DNA is yet unclear, the study found that women with more male DNA in their brains were less likely to suffer from Alzheimer’s disease, suggesting that male DNA could help protect mothers from this disease.
Researchers published their findings in the journal PLoS ONE. During pregnancy, mammal mothers and fetuses exchange DNA as well as cells. Previous work has shown that fetal cells can linger in a mother’s blood and bone for decades. This is called fetal microchimerism. This lingering fetal DNA could benefit the mother’s health, by promoting tissue repair and improving the immune system, but it may also cause adverse effects, such as autoimmune reactions.
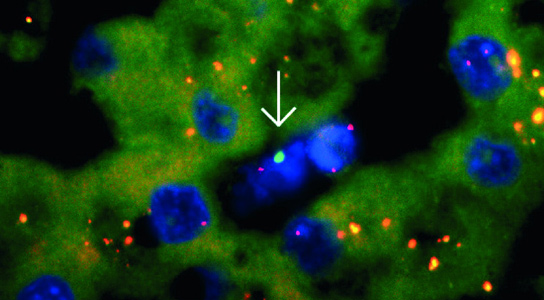
Scientists had previously shown that fetal microchimerism occurs in mouse brains, but had not yet discovered it in humans. A team of researchers, led by autoimmunity specialist and rheumatologist J. Lee Nelson, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, took samples from autopsied brains of 59 women who died between the ages of 32 and 101.
They found evidence of male DNA in the brains of 63% of the women. The DNA was scattered across multiple brain regions. The team also examined the brains for signs of Alzheimer’s in order to correlate the disease and the observed microchimerism. 33 had AD, but the women with AD had significantly less male DNA in their brains than the ones who didn’t have it.
It remains unclear whether that correlation implies that fetal male DNA helps protect women against AD and it’s difficult to reach any firm conclusions between microchimerism and AD because the team had little information about the pregnancies of the women in their study.
Reference: “Male Microchimerism in the Human Female Brain” by William F. N. Chan, Cécile Gurnot, Thomas J. Montine, Joshua A. Sonnen, Katherine A. Guthrie and J. Lee Nels, 26 September 2012, PLoS ONE.
DOI: 10.1371/journal.pone.0045592
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
“Bearing a Male Child Can Alter a Woman’s Brain” — especially about 15 years after the birth!
fetal microchimerism transporting male DNA from fetus to mother`s brain is talked about in this article. If the lingering male DNA in the brain causes delayed onset of Alzheimers Disease in women then there should be more women than men with AD isn`t it? The fact is otherwise with men having their own male DNA suffering from AD more than women.So, it remains unclear for the relationship between male DNA and AD and no firm conclusions can be arrived at. Thank You.
Need no further proof then mudsharks!
Why would a person choose to take on a more disadvantageous if not primitive and violent culture and lifestyle? If you have ever met them . They b some Crazy A$$ Dumb Biatches! 100