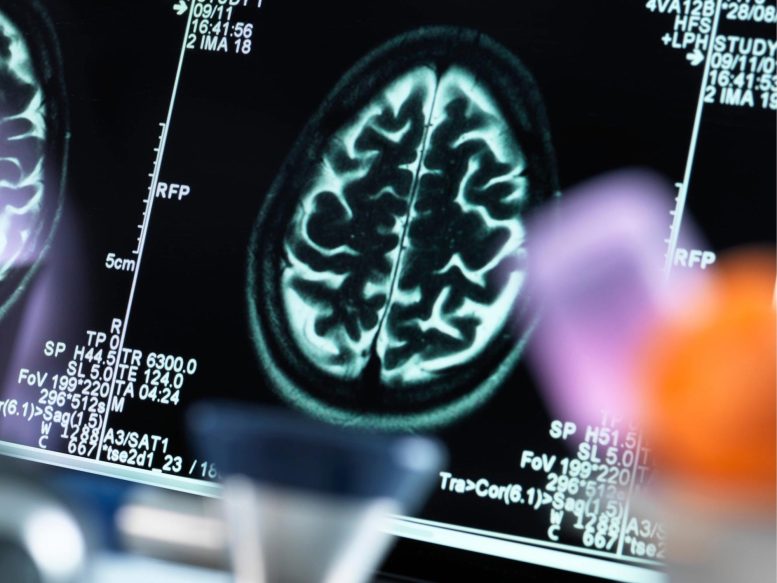
Brain Scan
At 11:00 am ET today (November 1, 2021), the results will be released for research that studied Incidence of blood clot in brain after Johnson & Johnson/Janssen COVID-19 vaccination.
What The Study Did: Resarchers compared post-Ad26.COV2.S (Johnson & Johnson/Janssen) vaccination cerebral venous sinus thrombosis (CVST, a blood clot in the brain) rates with prepandemic rates to estimate postvaccination CVST risk. The rate of this rare adverse effect must be considered in the context of the effectiveness of the vaccine in preventing COVID-19.
Authors: Aneel A. Ashrani, M.D., M.S., of the Mayo Clinic in Rochester, Minnesota, is the corresponding author.
Link: JAMA Internal Medicine
The study found a “significantly higher” brain blood clot (cerebral venous sinus thrombosis / CVST) incidence rate in people after vaccination with the Johnson & Johnson COVID-19 vaccine. The highest risk was in women between the ages of 30 and 49, with most CVST events happening within 15 days of vaccination.
Although this is a serious side effect, the authors caution that “the higher rate of this rare adverse effect must be considered in the context of the effectiveness of the vaccine in preventing COVID-19.”

Reference: “Age- and Sex-Specific Incidence of Cerebral Venous Sinus Thrombosis Associated With Ad26.COV2.S COVID-19 Vaccination” by Aneel A. Ashrani, MD, MS; Daniel J. Crusan, BS; Tanya Petterson, MS; Kent Bailey, PhD and John A. Heit, MD, 1 November 2021, JAMA Internal Medicine.
DOI: 10.1001/jamainternmed.2021.6352








Hey……I thought they already did all the studying on the vaccines that needed to be done and that they are safe and everything is fine. Well, good thing their legal liability has been waived by the government.
No medication or medical treatment is perfectly safe for everyone and it is unreasonable to expect them to be. The operative guideline is, “the higher rate of this RARE adverse effect must be considered in the context of the effectiveness of the vaccine in preventing COVID-19.” For rare effects, samples much larger than trials have to be obtained for the problem to be observed. It is sort of like the opposite of winning the lottery. Lots of people play, but very few win.
Who are you to say what’s reasonable as per the safety of any medicine? I and I alone will decide what’s a reasonable risk for ME to accept and YOU can shove your opinion where the sun don’t shine.
I second your point of view JC. Everyone knows themselves best. Give people the information and let them decide. What is so hard about that?
The ongoing COVID-19 nonsense here in the United States exists solely and exclusively because our governments have failed to use the correct treatment. They used so-called “vaccines” when Japan has just proven, in less than ONE MONTH, that Ivermectin can wipe out the disease. IVM was awarded the Nobel prize for medicine in 2015. One of the 3 most important drugs in human history: Aspirin, Penicillin, and Ivermectin. Get your Ivermectin today while you still can!
Nice try bot and/or troll. If your mindless comments weren’t enough give you away, your selected profile name surely did. If not a bot, you’re a mindless parrot.
Lmfao. You just believe whatever you read on Facebook. Japan has 70% of their population vaccinated and they don’t cry and wine over wearing mask. They know from previous epidemics that mask work and they aren’t fighting their government over mitigation. Japan is doing better than America because they are smarter, not because of ivermectin. Damn, you people need better sources.
I wouldn’t get the vaccine and risk my life for it. You never know if you are the one to get blood clots. Stop spreading fear of Covid and come up with treatments! The vaccines don’t work anyway! The government is keeping doctors from using treatments that they know work!
Right on! When it’s been proven safe over five years, then I’ll THINK about getting it. But honestly, I’d rather take my chances with nature.
Just do us a favor and foot the bill while you’re ‘taking your chances’ then we’re all good.
Don’t you ever get tired of being proved wrong?
Right…only if it’s you that dies of a brain hemorrhage…this rare side effect kills …and all for a virus that’s 98.9% recoverable.
Insanity!!
Well, when your legal liability has been waived by the government, you can say it is safe. How many of these companies should dare say it’s safe if they didn’t have their legal liability waiver?
ALL vaccines carry some risks,this is but one of the many adverse complications associated with vaccines, antibiotics, etc.
Right. It’s safe. Let’s pump it in to everyone including our babies. All the testing has been done. Damn people. I was told by the msm months ago that the stories of blood clotting was just conspiracy theories. Screw you msm I hope the lot of you get taken down by these vaccines. Safe and effective my ass. And then the fully vaccinated are still spreading it. Wtf
I agree with u Terry
I work in a nursing home and I have several residents long term that came here because of the side effects from the vaccine.one has a brain bleed.one confine to wheelchair,family tells me they were fine till they took the vaccine.The nursing home has now mandated that I get the vaccine by January 11th, Screw them I aint taking it so I guess I will loose my job.my life is more important My family is not taking it either. We got to stand up and be strong
While these soon doctors try to highlight RARE, they are forgetting that none of the “vaccines” prevent Covid as they state several times in the articles.
To all those people who think being vaccinated and ‘saving lives’ is such a good thing, have you no consideration for generations to come, YOUR CHILDREN, GRANDCHILDREN AND THEIR SUCCESSORS. This planet is overcrowded. The planet has its own way of regulating the population to keep it in equilibrium either by wars or pandemics. If it is not Covid 19 that reduces the population then starvation, water shortage and wars over lack of space the grow enough food will. If you think you are so good saving lives now you are just selfish. This thing needs to take its natural course to reduce the population for the good of those that follow.
Was on Blood thinners for quite a few years and had no problem taking it along with Diet my blood work was perfect every month until I got the Covid Vaccine My blood became thick
I really don’t believe the context is going to make a hill of beans to those who end up with thus particular side effect. They’re more than likely already pushing up daisies.
My Nephew had the vaxcine and died of an aneurysm, these A holes van say all they want but if it were their relitive it would be a completely diffent story.
My husband passed away mid September, from complications of CVST. He received the Johnson and Johnson vaccine in mid May.
I believe the Johnson and Johnson vaccine killed my husband.
We should not be forced into taking any vaccine.
WRONG! It is NOT unreasonable expect a pure, no complications “vaccine” Especially if people are being forced to get it in this draconian gov, or lose their jobs!
I was NOT a anti-vaxer until now! Until my eyes opened to the evil these elite globalists are doing and I see how Vaccines have caused so much pain, diseases, autoimmune issues and in many cases, have killed people.
Do you wonder why the human race appears that it’s getting sicker and weaker? Look into vaccines that are administered by pharmaceuticals. Look who profits from you when you get sick and need endless medical help and drugs.
I dare you to post this… Or, have you confirmed what I said above and we are we no longer in free/fair country with a First Amendment?!?
Bill Gates talk about how they plan on lowering the population on TED TALKS VIDEO. He briefly states through vaccines and healthcare they will lower the population…
And, look up the George guidestones which are pagan commandments one states to keep the population under five hundred million people… The vaccines ARE being used to kill people!
Also, the nurses are being told NOT to pull back on the plunger when administering the IM vax. But if they don’t pull back to make sure it’s not in a vein, then it could be fatal!
I am sure that the person with the clot and their families are comforted by the thought that they are the sacrificial lamb “for the greater good”
And, of course, no liability or recourse for the “freely chosen” allowing of being vaccinated.
Wow, this really shows how ignorant people are in today’s society. If you think for one minute that this vaccination roll out is not safe to the general public, or that the infection itself (COVID-19) is population control, or any other, amongst numerous, conspiracy theories out there, than you obviously don’t understand statistics, medical science, unexplained illness effects, world population growth, pandemics, or anything else any of you think this has to do with. If a side effect from a vaccine occurs in less than 10 percent of those given to but helps 70 percent of people from the infection becoming life threatening, and from that 10 percent that a side effect occurs only 10 percent of them actually result in death, that’s a mere 1 percent of those given actually die from it. But 70 percent it helps leaving 20 percent unaffected, didn’t help but didn’t hurt. I would say that’s pretty good odds considering the odds of not getting the infection if you do nothing and carry on normally. These percent numbers are not exactly but just an example to get the idea. Also I am not vaccinated but my close family is. Personally I have a death wish because I believe we have failed in humanity as a whole and think it’s about time for the one(s) in charge of this thing called life to hit the reset button and clear the slate, let us try again to be better and do better as humans. Evolve to a greater good that lives for the good of life everyday. Not to the negative war driven greedy selfish idiots that we seemed to have taken our society to. Corruption beyond a doubt, unforgivable criminal acts by the thousands daily, what we consume, what we watch, what we are taught, what we want and desire, the things we think, how we treat each other, etc, exactly. The daily devil’s that disturb destiny with destruction result in detrimental devastation. While those peaceful providence of protected people who portray positive passages result in powerful places of impressions. Power to the people.
So if you do the math, this “signicantly higher” risk equals 0.0000032184% of people getting just that particular vaccine. Do your research people, these guys use unnecessary inflamitory language to spark clicks and engagement.