A redesigned catalyst appears to sidestep a major bottleneck in CO2-to-methanol conversion by separating where key reaction steps occur.
Efficient methanol production could play an important role in carbon recycling, turning captured carbon dioxide (CO2) into a useful chemical feedstock and fuel ingredient. In principle, the chemistry works best at low temperatures, where converting CO2 into methanol is thermodynamically favorable. In practice, though, there is a major obstacle: CO2 is hard to activate under those milder conditions, so catalysts tend to perform poorly.
Turning up the heat helps the reaction move faster, but it creates another problem. Higher temperatures also encourage the reverse water gas shift reaction, which diverts CO2 toward carbon monoxide instead of methanol. That leaves researchers stuck with a familiar trade-off.
Conditions that improve activity often hurt selectivity, and conditions that favor selectivity often reduce output. This balancing act has been a major barrier to boosting methanol yield.
A New Catalyst Design Strategy
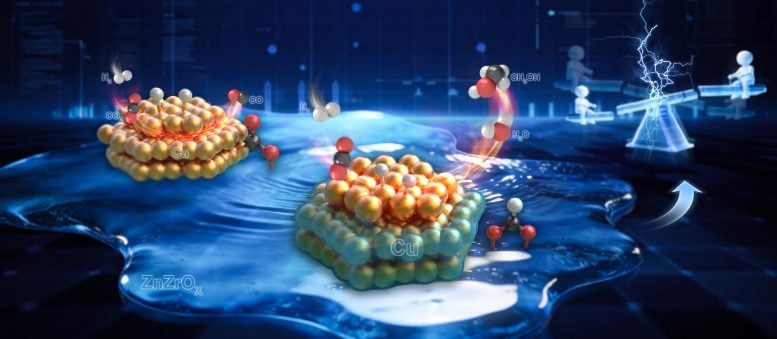
In a study published in Chem, a team led by Prof. Jian Sun and Prof. Jiafeng Yu at the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) introduced a new catalyst design. Their approach separates active sites in space using a strong metal support interaction (SMSI) driven overlayer structure, improving the efficiency of methanol production from CO2.
The team reshaped the catalyst surface and altered how reactants attach and break apart, as well as how the reaction proceeds. Under conditions of 300 ℃ (572 °F) and 3 MPa (about 435 psi), the system reached a space time yield of 1.2 g·gcat-1·h-1. This performance is roughly three times higher than that of standard commercial Cu/Zn/Al catalysts.
How the Reaction Pathway Changes
The researchers discovered that their design directs CO2 to adsorb and activate mainly on zirconia (ZrO2), steering the process toward methanol formation through the formate pathway.
This differs from the usual mechanism on copper sites, where the C=O bond is broken before hydrogenation. In the new system, hydrogenation happens first on ZrO2, followed by cleavage of the C=O bond. This shift reduces the formation of unwanted CO while maintaining the strong ability of copper sites to split H2.
“Our study may provide a new pathway to addressing the long-standing trade-off between activity and selectivity in methanol synthesis from CO2,” said Prof. Sun.
Reference: “Disentangling the activity-selectivity trade-off in CO2 hydrogenation to methanol” by Habib Zada, Jiafeng Yu, Chuanyan Fang and Jian Sun, 13 March 2026, Chem.
DOI: 10.1016/j.chempr.2026.102942
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
CO2 gas can generate electricity when ionized and it is passed through a conductive material and without any ignition source. See U.S. Patent 3,022,430 by T.T. Brown.
Were goin back to the future again GET IN
“This performance is roughly three times higher than that of standard commercial Cu/Zn/Al catalysts.”
So, three times what?
thanks for this