Scientists have uncovered a surprising overlap between cancer genetics and Alzheimer’s disease, involving mutations in the brain’s immune cells.
As we age, our cells quietly collect genetic mutations—most of them harmless, some potentially dangerous. But what if the same kinds of mutations that fuel cancer are also shaping diseases of the brain?
A new study from Boston Children’s Hospital, published in Cell, reveals that microglia, the brain’s resident immune cells, accumulate cancer-linked mutations, not to form tumors, but possibly to drive Alzheimer’s disease.
The research was led by Christopher Walsh, MD, PhD, Chief of the Division of Genetics and Genomics at Boston Children’s and an Investigator at the Howard Hughes Medical Institute. His colleagues included Alice Eunjung Lee, PhD, and August Yue Huang, PhD, also from the Division of Genetics and Genomics. All are professors at Harvard Medical School and associate members of the Broad Institute of MIT and Harvard. The team believes their findings could point to new ways to diagnose and treat Alzheimer’s disease.
“We find that to some extent, Alzheimer’s disease is a little like cancer—driven by the same mutations that drive blood cancers like lymphoma and leukemia,” said Walsh. “This is helpful because we have a lot of drugs to fight cancer, and some of them might be useful therapeutically for Alzheimer’s disease.”
Cancer-Linked Mutations Found in Alzheimer’s Brains
To investigate, the researchers analyzed 149 cancer-associated genes in brain tissue from 190 people with Alzheimer’s disease and compared the results with samples from 121 individuals without the condition. The Alzheimer’s samples showed a higher number of single-letter DNA changes, with many recurring in the same five cancer-related genes. This pattern indicates that microglia were accumulating mutations in specific targets.
Microglia act as the brain’s cleanup crew, removing debris as well as damaged or infected cells. Unlike most immune cells that travel through the bloodstream, microglia have long been thought to remain confined within the brain.
Many of the mutations identified in microglia are also common in blood cancers. Based on this overlap, the researchers examined blood samples from people with Alzheimer’s disease to see if the same mutations were present. They did not expect to find them, but the results showed that blood cells from these patients carried the same cancer-related mutations.
Surprising Discovery: Blood Cells Carry Brain Mutations
“It was actually a really unexpected finding that suggests a totally new mechanism for Alzheimer’s disease pathogenesis,” said Huang. “The findings mean that the blood’s immune cells with cancer mutations are likely getting into the brain and contributing to disease.”
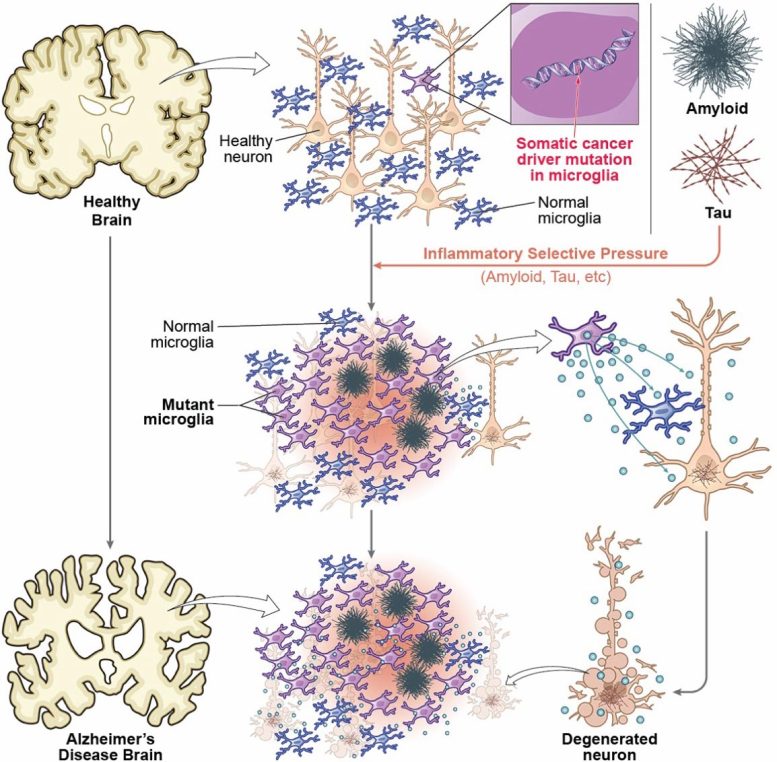
The team proposes that the blood-brain barrier may become less effective with age or injury, allowing immune cells from the bloodstream to enter the brain. Once inside, these cells may take on characteristics similar to microglia. At the same time, protein buildup in the brain can trigger microglia to multiply and react.
Cells with a growth advantage, such as those carrying cancer-related mutations, may come to dominate. These altered cells can create a more inflammatory environment, which may damage nearby neurons and contribute to Alzheimer’s disease.
Implications for Diagnosis and Future Research
“Because it’s hard to access brain tissue in a living patient, genetic screens using blood samples could be developed to test whether a person carries these mutations and has an increased risk of developing Alzheimer’s disease,” said Lee.
In a follow-up study posted as a preprint on bioRxiv, Huang and Lee found that cancer-related mutations detected in blood samples were linked to a higher risk of Alzheimer’s disease, even when accounting for APOE4, a well-known genetic risk factor.
References:
“Somatic cancer variants enriched in Alzheimer’s disease microglia-like cells drive inflammatory and proliferative states” by August Yue Huang, Zinan Zhou, Maya Talukdar, Liz Enyenihi, Michael B. Miller, Brian Chhouk, Ila Rosen, Mengyue Zheng, Minye Zhou, Averill Yang, Edward Stronge, Madel Durens, Minh Nguyen, Jaejoon Choi, Boxun Zhao, Sattar Khoshkhoo, Junho Kim, Rebecca Andersen, Zheming An, Yuchen Cheng and Christopher A. Walsh, 21 April 2026, Cell.
DOI: 10.1016/j.cell.2026.03.040
“Clonal Hematopoiesis Mutations Increase Risk of Alzheimer’s Disease with APOE ε3/ε3 Genotype” by Jaejoon Choi, Kyung Sun Park, Yann Le Guen, Jong-Ho Park, Zinan Zhou, Liz Enyenihi, Ila Rosen, Yoon-Ho Choi, Christopher A. Walsh, Jong-Won Kim, August Yue Huang and Eunjung Alice Lee, 21 May, 2025, bioRxiv.
DOI: 10.1101/2025.05.19.654981
This work was done in collaboration with the Icahn School of Medicine at Mount Sinai and was supported by the Howard Hughes Medical Institute, the National Institute on Aging, the NIH Common Fund through the Somatic Mosaicism Across Human Tissues (SMaHT) consortium, and the Suh Kyungbae Foundation (SUHF).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.