Scientists just uncovered the cellular “blueprint” that could one day let us regrow real teeth.
Researchers at Science Tokyo have uncovered two distinct stem cell lineages that play a central role in forming tooth roots and the surrounding alveolar bone. By studying genetically modified mice and tracking how individual cells develop over time, the team revealed key signaling processes that guide how stem cells specialize during tooth growth. The findings could help lay the groundwork for future treatments aimed at regenerating teeth and supporting bone.
Why Regrowing Teeth Has Been So Difficult
Replacing lost teeth has long relied on artificial solutions like implants and dentures. While effective, these options cannot fully match the structure, function, or natural feel of real teeth. For years, scientists have been working to understand how teeth form in the first place, hoping to unlock ways to regrow them naturally.
That challenge is complex. Tooth development depends on tightly coordinated interactions between multiple cell types and tissues, including dental pulp, the enamel organ, and bone-forming cells in the jaw. These components communicate through intricate signaling networks that carefully control how each part of the tooth, from crown to root, takes shape. Despite decades of research, many details of this process remain unclear.
Tracking Stem Cells in Developing Teeth
To close these knowledge gaps, a team led by Assistant Professor Mizuki Nagata from the Department of Periodontology, Graduate School of Medical and Dental Sciences at Institute of Science Tokyo (Science Tokyo), Japan, and Dr. Wanida Ono of the University of Texas Health Science Center at Houston (UTHealth), USA, worked with collaborators from the University of Michigan, USA, and other institutions. They conducted two related studies on how stem cells differentiate during tooth development. The results were published in Nature Communications.
Using genetically engineered mice along with advanced lineage tracing methods, the researchers followed how cells behave at the ‘tip’ (apical region) of growing tooth roots. Techniques such as high-resolution microscopy, fluorescent labeling, and gene silencing allowed them to observe how specific signaling proteins influence what types of cells stem cells ultimately become.
Two Distinct Stem Cell Lineages Discovered
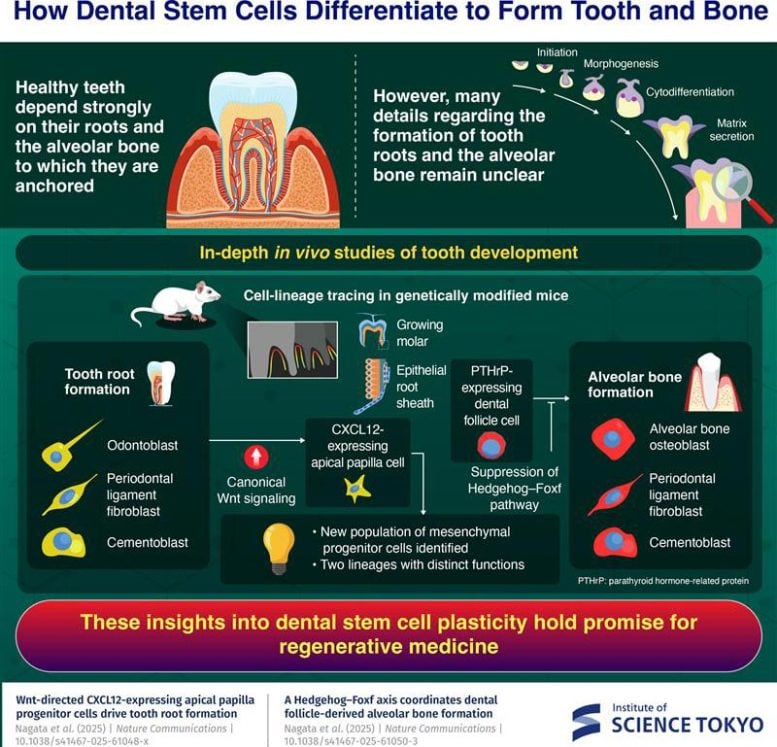
The team identified a previously unknown group of mesenchymal stem cells that split into two separate developmental paths. One lineage is closely tied to the formation of the tooth root, while the other contributes to building the alveolar bone that anchors the tooth in place.
The first lineage originates in the apical papilla, a soft tissue region located within the epithelial root sheath at the tip of a developing tooth root. These cells produce CXCL12, a protein known for its role in bone formation in bone marrow. Through a signaling route called the canonical Wnt pathway, these CXCL12-expressing cells can become several different cell types. They can form odontoblasts, which create dentin in teeth, as well as cementoblasts that produce the outer layer of the root. Under regenerative conditions, they can even develop into osteoblasts that generate alveolar bone.
A Second Pathway Controls Bone Formation
The second lineage is found in the dental follicle, a sac-like structure that surrounds the developing tooth and helps form the tissues that support it. Within this region, the researchers identified cells that express parathyroid hormone-related protein (PTHrP). These cells are capable of differentiating into cementoblasts, ligament fibroblasts, and osteoblasts that form alveolar bone.
However, this transformation is not automatic. It depends on specific molecular conditions. As Nagata explains, “We observed that the Hedgehog–Foxf pathway needs to be suppressed to drive the alveolar bone osteoblast fate of PTHrP-expressing cells in the dental follicle, unraveling a unique tooth-specific mechanism of bone formation requiring deliberate on–off regulation of Hedgehog signaling.”
A Step Toward Regenerating Teeth and Bone
Together, these discoveries provide a clearer picture of how teeth and their supporting bone develop inside the body. By mapping the roles of these two stem cell lineages and the signals that guide them, researchers now have a stronger framework for understanding tooth root formation.
Nagata highlights the broader impact, stating, “Our findings provide a mechanistic framework for tooth root formation and pave the way for innovative stem-cell-based regenerative therapies for dental pulp, periodontal tissues, and bone.”
The studies were published under the titles “Wnt-directed CXCL12-expressing apical papilla progenitor cells drive tooth root formation” and “A Hedgehog–Foxf axis coordinates dental follicle-derived alveolar bone formation.”
References:
“Wnt-directed CXCL12-expressing apical papilla progenitor cells drive tooth root formation” by Mizuki Nagata, Gaurav T. Gadhvi, Taishi Komori, Yuki Arai, Hiroaki Manabe, Angel Ka Yan Chu, Ramandeep Kaur, Meer Ali, Yuntao Yang, Chiaki Tsutsumi-Arai, Yuta Nakai, Yuki Matsushita, Nicha Tokavanich, W. Jim Zheng, Joshua D. Welch, Noriaki Ono and Wanida Ono, 1 July 2025, Nature Communications.
DOI: 10.1038/s41467-025-61048-x
“A Hedgehog–Foxf axis coordinates dental follicle-derived alveolar bone formation” by Mizuki Nagata, Gaurav T. Gadhvi, Taishi Komori, Yuki Arai, Chiaki Tsutsumi-Arai, Angel Ka Yan Chu, Seth N. Nye, Yuntao Yang, Shion Orikasa, Akira Takahashi, Peter Carlsson, W. Jim Zheng, Joshua D. Welch, Noriaki Ono and Wanida Ono, 2 July 2025, Nature Communications.
DOI: 10.1038/s41467-025-61050-3
Funding: National Institutes of Health, Cancer Prevention and Research Institute of Texas, Japanese Society of Periodontology, Japan Society for the Promotion of Science
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
I have been reading about this stem cell for over 5 years but no one is really anticipate in having it done. I like to know what is the procedure? I have this done to others and me also why is it good thing? i’m not being anything done to help people grow new teeth if you if you have any patience or how anytime to get back with me and tell me how can we have it done? Have a great day in May you be safe
‘I’m been reborn again’