
Scientists have developed a method to alter the color and brightness of rare earth element luminescence by changing their chemical environment, enabling the design of advanced light-emitting materials.
Researchers at HSE University and the Institute of Petrochemical Synthesis of the Russian Academy of Sciences have discovered a method to control both the color and brightness of light emitted by rare earth elements. Typically, the luminescence of these elements is highly predictable. For example, cerium ions usually emit light in the ultraviolet range.
However, the scientists demonstrated that this emission can be altered. By creating a specific chemical environment, they caused a cerium ion to emit a yellow glow instead of its usual ultraviolet light. These findings could lead to the development of advanced light sources, display technologies, and laser systems. The study was published in Optical Materials.
Rare earth elements are important in microelectronics, LEDs, and fluorescent materials because they can emit light in precisely defined colors. This ability results from the behavior of their electrons when they absorb and release energy.
When an atom absorbs energy from a source such as light or an electric current, one of its electrons can move to a higher energy level. This excited state is unstable. After a short time, the electron returns to its original energy level and releases the excess energy in the form of light. This process is known as luminescence.
Orbital Transitions and Their Typical Stability
In rare earth elements, the glow results from electron transitions between 4f orbitals—regions around the atomic nucleus where electrons can reside. Typically, the energy of these transitions is fixed, meaning the color of the glow remains constant: cerium emits invisible ultraviolet light, while terbium emits green.
The 4f orbitals are situated deep within the atom and interact minimally with the surrounding environment. In contrast, the 5d orbitals are sensitive to external influences but generally do not contribute to the luminescence of lanthanides due to their excessively high energy.
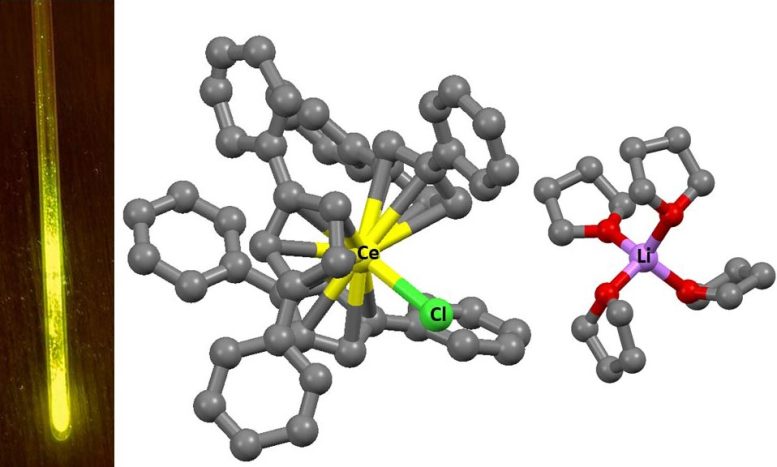
However, scientists from HSE University and the Institute of Petrochemical Synthesis of the Russian Academy of Sciences have demonstrated that the color of the radiation can be altered by adjusting the chemical environment of the metals. They synthesised cerium, praseodymium, and terbium complexes using organic ligands—molecules that surround metal ions. These ligands shape the geometry of the complex and influence its properties. In all cases, three cyclopentadienyl anions were symmetrically arranged around the metal.
These anions consist of regular pentagons of carbon atoms, to which large organic fragments are attached, providing the required structure for the complex. This environment generates a specific electrostatic field around the ion, which alters the energy of the 5d orbitals and, consequently, affects the luminescence spectrum.
Unveiling the Mechanism Behind the Change
“Previously, a change in the color of the glow had been observed, but the underlying mechanism was not understood. Now, in collaboration with our physicist colleagues, we have been able to understand the mechanism behind this effect. We deliberately designed compounds with an electronic structure that is atypical for lanthanides. Rather than focusing on a single example, we synthesised a series of compounds from cerium to terbium to observe how their properties change and to identify common patterns,” comments Daniil Bardonov, a master’s student at the HSE Faculty of Chemistry.
In conventional compounds, cerium emits ultraviolet light with wavelengths between 300 and 400 nanometres. In the new complexes, its emission shifted to the red range, reaching up to 655 nanometres. This indicates that the energy gap between the 4f and 5d levels has decreased. A similar rearrangement of electronic levels was observed in the other lanthanides studied, also resulting in changes to their luminescence.
“To understand how this process works, it’s important to first grasp the mechanism of energy transfer. Typically, a ligand molecule absorbs ultraviolet light, enters an excited state, and then transfers this energy to the metal atom, causing it to emit light,” explains Dmitrii Roitershtein, Academic Supervisor of the Chemistry of Molecular Systems and Materials Programme and co-author of the paper. “However, in the new compounds, the process occurred differently: energy was transferred not directly to the 4f electrons, but via an intermediate 5d state.”
The researchers believe that being able to predict the luminescence spectrum will make it possible to design materials with desired properties more efficiently by eliminating the need for time-consuming trial and error. This could facilitate the creation of new and advanced light sources.
“We were able to demonstrate exactly how the environment of an atom influences its electronic transitions and lanthanide luminescence,” says Fyodor Chernenkiy, a bachelor’s student at the HSE Faculty of Chemistry. “We can now intentionally select the structure of compounds to control luminescence and produce materials with specific optical properties.”
Reference: “Efficiency of the energy transfer through 5d state of the Ln3+ ion in complexes with diarylcyclopentadienyl ligands” by Lada N. Puntus, Evgenia A. Varaksina, Daniil A. Bardonov, Mikhail E. Minyaev, Konstantin A. Lyssenko, Theodor A. Chernenkiy, Ilya V. Taydakov, Ilya E. Nifant’ev and Dmitrii M. Roitershtein, 27 January 2025, Optical Materials.
DOI: 10.1016/j.optmat.2025.116734
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
I like it
Your parents would’ve wanted a comment here. Please and thank you.
I like it
I have study of competitive exams
If someone says they got a God particle