Ever since it was proposed that atoms are building blocks of the world, scientists have been trying to understand how and why they bond to each other. Be it a molecule (which is a group of atoms joined together in a particular fashion), or a block of material or a whole living organism, ultimately, everything is controlled by the way atoms bond, and the way bonds break.
The challenge is that lengths of chemical bonds are between 0.1 – 0.3 nm, about half a million times smaller than the width of a human hair, making direct imaging of bonding between a pair of atoms difficult. Advanced microscopy methods, such as atomic force microscopy (AFM) or scanning tunneling microscopy (STM), can resolve atomic positions and measure bond lengths directly, but filming chemical bonds to break or to form, with spatiotemporal continuity, in real time, still remains one of the greatest challenges of science.
This challenge has been met by a research team from the UK and Germany led by Professor Ute Kaiser, head of the Electron Microscopy of Materials Science in the University of Ulm, and Professor Andrei Khlobystov in the School of Chemistry at the University of Nottingham they have published ‘Imaging an unsupported metal-metal bond in dirhenium molecules at the atomic scale’ in Science Advances, a journal of the American Association for the Advancement of Science covering all aspects of scientific endeavor.
Atoms in a nano test tube
This group of researchers are known for their pioneering use of transmission electron microscopy (TEM) to film ‘movies’ of chemical reactions at the single-molecule level, and dynamics of tiny clusters of metal atoms in nanocatalysts utilize carbon nanotubes — atomically thin hollow cylinders of carbon with diameters at the molecular scale (1-2 nm) as miniature test tubes for atoms.
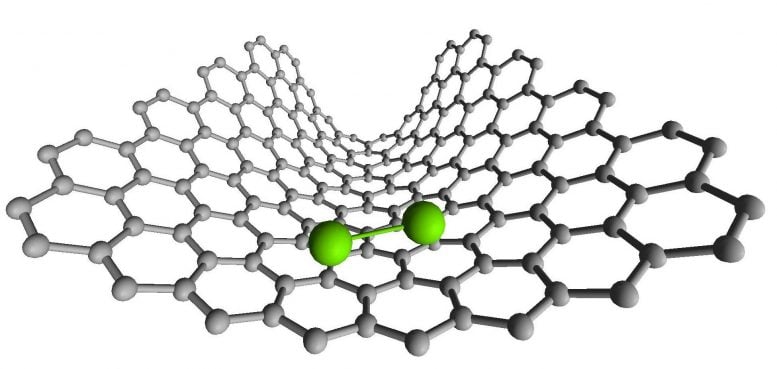
Professor Andrei Khlobystov, said: “Nanotubes help us to catch atoms or molecules, and to position them exactly where we want. In this case, we trapped a pair of rhenium (Re) atoms bonded together to form Re2. Because rhenium has a high atomic number it is easier to see in TEM than lighter elements, allowing us to identify each metal atom as a dark dot.”
Professor Ute Kaiser added: “As we imaged these diatomic molecules by the state of the art chromatic and spherical aberration-corrected SALVE TEM, we observed the atomic-scale dynamics of Re2 adsorbed on the graphitic lattice of the nanotube and discovered that the bond length changes in Re2 in a series of discrete steps.”
A dual use of electron beam
The group have a rich track record of using an electron beam as a tool for dual-purpose: precise imaging of atomic positions and activation of chemical reactions due to energy transferred from fast electrons of the electron beam to the atoms. The “two-in-one” trick with TEM allowed these researchers to record movies of molecules reacting in the past, and now they were able to film two atoms bonded together in Re2 ‘walking’ along the nanotube in a continuous video. Dr. Kecheng Cao, Research Assistant at Ulm University who discovered this phenomenon and performed the imaging experiments, said: ‘It was surprisingly clear how the two atoms move in pairs, clearly indicating a bond between them. Importantly, as Re2 moves down the nanotube, the bond length changes, indicating that the bond becomes stronger or weaker depending on the environment around the atoms.’
Breaking the bond
After a period of time, atoms of Re2 exhibited vibrations distorting their circular shapes onto ellipses and stretching the bond. As the bond length reached a value exceeding the sum of atomic radii, the bond snapped and vibration ceased, indicating that the atoms became independent of one another. A little later the atoms joined together again, reforming a Re2 molecule.
Dr. Stephen Skowron, Postdoctoral Research Assistant at University of Nottingham who carried out the calculations for Re2 bonding, said: ‘Bonds between metal atoms are very important in chemistry, particularly for understanding magnetic, electronic, or catalytic properties of materials. What makes it challenging is that transition metals, such as Re, can form bonds of different order, from single to quintuple bonds. In this TEM experiment, we observed that the two rhenium atoms are bonded mainly through a quadruple bond, providing new fundamental insights into transition metal chemistry’.
Electron microscope as a new analytical tool for chemists
Andrei Khlobystov, said: “To our knowledge, this is the first time when bond evolution, breaking, and formation was recorded on film at the atomic scale. Electron microscopy is already becoming an analytical tool for determining structures of molecules, particularly with the advance of the cryogenic TEM recognized by 2017 Nobel Prize in Chemistry. We are now pushing the frontiers of molecule imaging beyond simple structural analysis, and towards understanding dynamics of individual molecules in real time.” The team believe that one day in the future electron microscopy may become a general method for studying chemical reactions, similar to spectroscopic methods widely used in chemistry labs.
Reference: “Imaging an unsupported metal–metal bond in dirhenium molecules at the atomic scale” by Kecheng Cao, Stephen T. Skowron, Johannes Biskupek, Craig T. Stoppiello, Christopher Leist, Elena Besley, Andrei N. Khlobystov and Ute Kaiser, 17 January 2020, Science Advances.
DOI: 10.1126/sciadv.aay5849
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.