
A team from IBM in Zürich has released images they captured of single molecules that are so detailed that the type of atomic bonds between their atoms can be discerned.
The scientists published their findings in the journal Science. The same IBM team took the first ever single-molecule images in 2009 and most recently published the images of the pentacene, the molecule shaped like the Olympic rings.

These new images will allow scientists to study the imperfections of graphene or plotting where electrons go during chemical reactions.
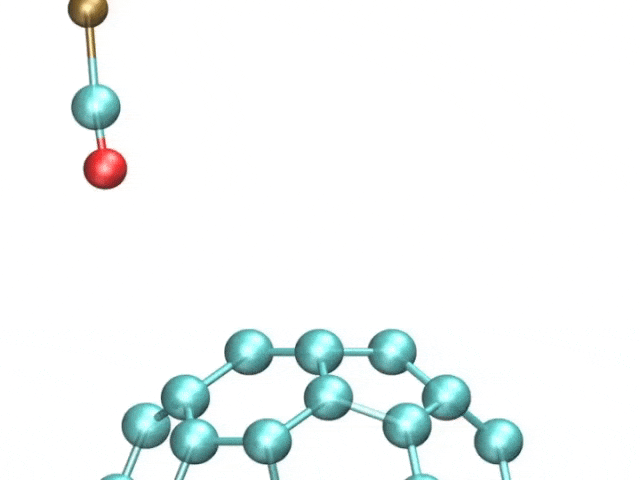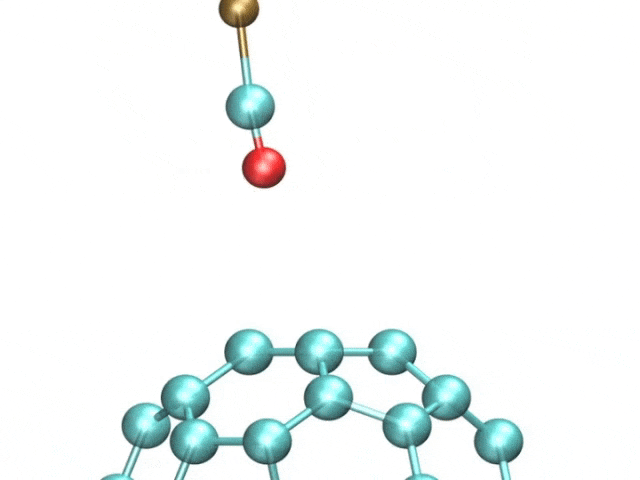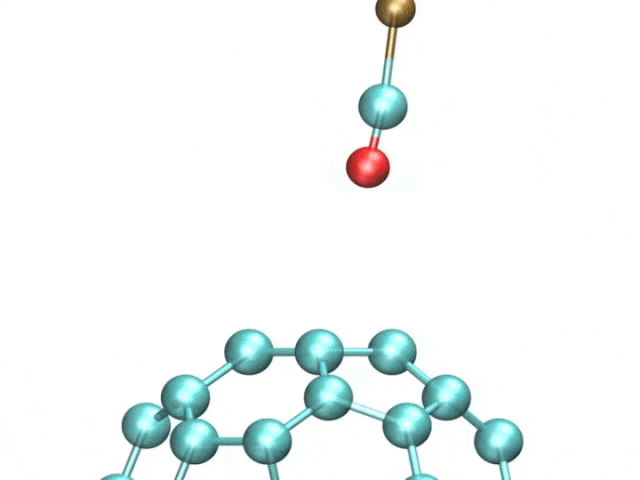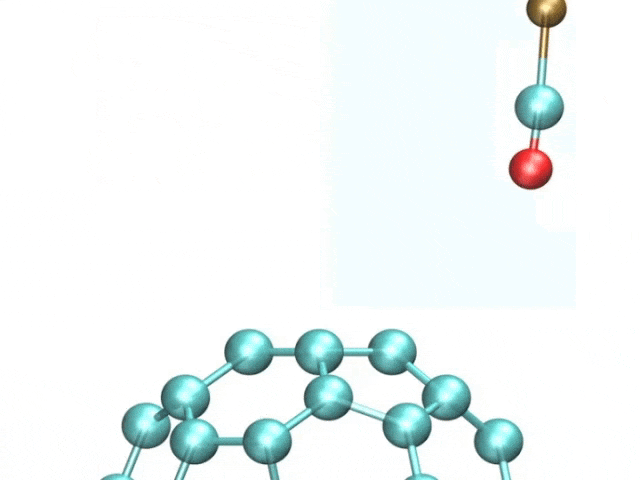
The team included French and Spanish collaborators, and they used a variant of a technique called atomic force microscopy (AFM). AFM uses a tiny metal tip that passes over a surface, where even tinier deflections are measured as the tip is scanned as it moves to and fro over a sample.
The pentacene molecule was scanned by using a carbon monoxide molecule, which acted like a recording needle, allowing for unprecedented accuracy of the surfaces of atoms.
The experiments themselves must be isolated from any kind of vibrations. Room temperatures themselves induce movements of the AFM’s constituent molecules, which would blur the image, so the actual apparatus must be kept at -268 °C (-450 °F).
The tiny distortions in the regular hexagonal pattern of carbon bonds are what interested the researchers. Fullerenes and polyaromatic hydrocarbons, which have linked rings of carbon atoms at their cores, were the focus of this study.
The images showcased how long the atomic bonds are. The bright and dark spots correspond to higher and lower densities of electrons. There are different physical properties of different bonds.
Reference: “Bond-Order Discrimination by Atomic Force Microscopy” by Leo Gross, Fabian Mohn, Nikolaj Moll, Bruno Schuler, Alejandro Criado, Enrique Guitián, Diego Peña, André Gourdon and Gerhard Meyer, 14 September 2012, Science.
DOI: 10.1126/science.1225621
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.