
A new method for the easy production of vicinal diamines has been developed by chemists.
A team led by Professor Frank Glorius of the University of Münster has developed a new method for producing vicinal diamines, a common structure found in biologically active molecules, natural products, and drugs. These compounds contain two functional groups, each with a nitrogen atom bonded to two neighboring carbon atoms. The team’s method, which was published in the journal Nature Catalysis, is a direct and efficient way to produce these compounds.
The new method developed by the team at the University of Münster does not require the use of transition metals or iodine reagents as catalysts, unlike other methods. Instead, it utilizes light energy to produce the desired diamines from various electron-rich aromatic hydrocarbons, such as arenes and heteroarenes. This makes the process more efficient and effective than other methods.
“In this way, we obtain a series of vicinal diamines that were previously difficult to produce. In doing so, we can precisely control the sites where the functional groups are located,” explains first author Dr. Guangying Tan.
Regioselective Reaction Control
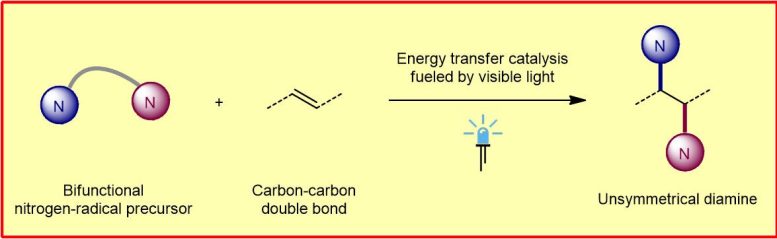
To this end, the chemists developed a class of special nitrogen radical precursors that simultaneously generate two nitrogen-centered radicals with different reactivities via an energy transfer process. By “regioselectively” adding two of these radicals stepwise via carbon-carbon double bonds, the scientists produce the unsymmetrically constructed vicinal diamines. “Regioselective” means that the reaction occurs at defined sites on the molecules. The functional groups (amino groups) can then be further modified. The fact that the diamines synthesized in this way are not symmetrical, in contrast to a symmetrical structure, opens up a much greater variety of functional groups to be considered.
“The molecules of life consist largely of carbon chains and rings of varying size and complexity. The decoration of these ‘plain’ chains with other elements is crucial for the resulting properties of these compounds,” Frank Glorius explains the background. A key role is played by the elements oxygen and nitrogen. Chemists refer to these non-carbon elements as heteroatoms.
“Methods for the efficient and controlled introduction of these heteroatoms into artificially produced, biologically active structures are therefore of great importance,” Frank Glorius emphasizes. “This also applies to the vicinal diamines we are focusing on.”
The chemists perform the diamination reaction under irradiation with blue light-emitting diodes (LEDs) and use an inexpensive and commercially available thioxanthone as an organic photosensitizer.
Reference: “Energy transfer-enabled unsymmetrical diamination using bifunctional nitrogen-radical precursors” by Guangying Tan, Mowpriya Das, Roman Kleinmans, Felix Katzenburg, Constantin Daniliuc and Frank Glorius, 8 December 2022, Nature Catalysis.
DOI: 10.1038/s41929-022-00883-3
The study was funded by the German Research Foundation, Alexander von Humboldt-Stiftung, and Fonds der Chemischen Industrie.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.