COVID-19 drug trials may fail due to late patient enrollment. Early recruitment could drastically improve results with fewer participants.
A new modeling study suggests that significant variation in virus dynamics from person to person may be a contributing factor to inconsistent findings reported in clinical trials for antiviral COVID-19 drugs. Recruiting trial participants shortly after symptoms begin could reduce the number of participants required to detect significant antiviral drug effects, according to Shoya Iwanami and Shingo Iwami of Nagoya University in Aichi, Japan, Keisuke Ejima of Indiana University, Indiana, USA, and colleagues, who present these findings in the open-access journal PLOS Medicine.
An effective antiviral drug for COVID-19 would have significant global health benefits. However, clinical trials that test candidate drugs have produced inconsistent results, perhaps due to flaws in the way the trials are designed.
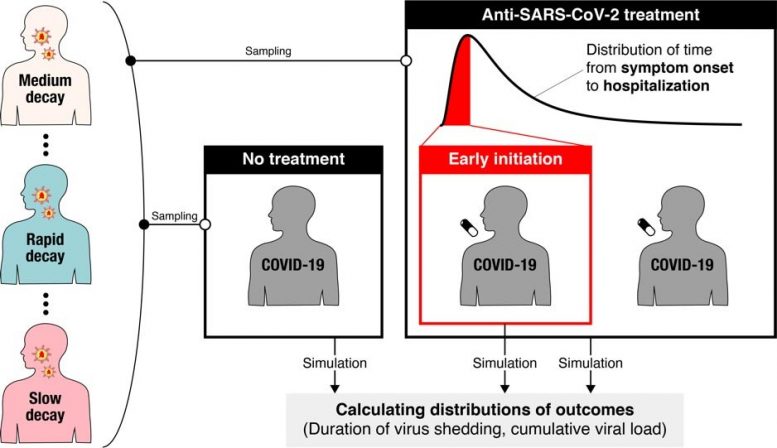
To address this issue, Iwanami and colleagues first utilized a model of the dynamics of SARS-CoV-2–the virus that causes COVID-19–once it has infected a person. They combined the model with clinical data to examine how viral load (the amount of virus in a person’s throat) changes over time, and found significant variation in the rate of decline between patients. These differences may contribute to the inconsistent results reported in non-randomized clinical trials so far.
Simulating Better COVID-19 Trial Strategies
Exploring further, the researchers simulated potential findings of randomized clinical trials for COVID-19 drugs that successfully interrupt virus replication. They found that, even if a drug reduced viral replication by 95%, the associated randomized clinical trial would need to enroll more than 13,000 people to receive the drug being tested, plus the same number of people to receive a placebo for comparison, in order to detect statistically significant differences in viral load. In most cases, those numbers would be unreasonably large.
However, when the researchers altered the simulated randomized clinical trials so that participants were treated within one day of onset of their symptoms, they found that only up to about 600 participants were needed for each group. This suggests that randomized clinical trials for COVID-19 drugs could be improved by enrolling participants as soon as possible after symptoms appear, or by setting enrolment criteria based on the time that has passed since symptom onset.
The researchers note that future studies could employ more detailed models of SARS-CoV-2 dynamics in order to produce more reliable calculations of the numbers of participants needed for randomized clinical trials to produce consistent results.
A New Approach to Trial Design and Drug Testing
“We found that if patients are recruited to clinical trials regardless of the time since symptom onset, the number should be over 10,000, which is unreasonably large,” Dr. Iwami adds. “This is because many patients are recruited too late to see antiviral treatment effect. Thus, we suggest recruiting only those who are still ‘new’ since symptom onset. If we recruit only patients in 2 days since symptom onset, only 500 patients need to be recruited. The approach we developed can be applied to other types of drugs and different infectious diseases. We are hoping to develop an online platform which supports designing clinical trials.”
Reference: “Detection of significant antiviral drug effects on COVID-19 with reasonable sample sizes in randomized controlled trials: A modeling study combined with clinical data” by Shoya Iwanami, Keisuke Ejima, Kwang Su Kim, Koji Noshita, Yasuhisa Fujita, Taiga Miyazaki, Shigeru Kohno, Yoshitsugu Miyazaki, Shimpei Morimoto, Shinji Nakaoka, Yoshiki Koizumi, Yusuke Asai, Kazuyuki Aihara, Koichi Watashi, Robin N. Thompson, Kenji Shibuya, Katsuhito Fujiu, Alan S. Perelson, Shingo Iwami and Takaji Wakita, 6 July 2021, PLOS Medicine.
DOI: 10.
Funding: This study was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers JP19J12319 (to S.Iwanami), JP18KT0018 (to S.I.), JP18H01139 (to S.I.), JP16H04845 (to S.I.), JP17H04085 (to K.W.), JP18K18146 (to K.E.), JP20H05042 (to S.I.), JP19H04839 (to S.I.), JP18H05103 (to S.I.); Japan Agency for Medical Research and Development (AMED) Grant Numbers JP19gm1310002 (to S.I.), JP20wm0325007h0001 (to S.I.), JP20wm0325004s0201 (to S.I.), JP20wm0325012s0301 (to S.I.), JP20wm0325015s0301 (to S.I.), JP19fk0410023s0101 (to S.I.), JP19fk0108050h0003 (to S.I.), JP19fk0108156h0001 (to S.I.), JP20fk0108140s0801 (to S.I.), JP20fk0108413s0301 (to S.I.), JP19fk0210036h0502 (to S.I.), JP19fk0210036j0002 (to K.W.), JP19fk0310114h0103 (to S.I.), JP19fk0310114j0003 (to K.W.), JP19fk0310101j1003 (to K.W.), JP19fk0310103j0203 (to K.W.); Japan Science and Technology Agency (JST) MIRAI (to S.I. and K.W.); Moonshot R&D Grant Number JPMJMS2021 (to K.A. and S.I.) and JPMJMS2025 (to S.I.); Mitsui Life Social Welfare Foundation (to S.I. and K.W.); Shin-Nihon of Advanced Medical Research (to S.I.); Suzuken Memorial Foundation (to S.I.); Life Science Foundation of Japan (to S.I.); SECOM Science and Technology Foundation (to S.I.); The Japan Prize Foundation (to S.I.); Daiwa Securities Health Foundation (to S.I.); The Yasuda Medical Foundation (to K.W.); Smoking Research Foundation (to K.W.); and The Takeda Science Foundation (to K.W.); NSF PHY-2031756 (to A.S.P.); NIH R01-OD011095 and R01-AI028433 (to A.S.P.); and Los Alamos National Laboratory LDRD Program (to A.S.P.); the MIDAS Coordination Center (MIDASSUGP2020-6) by a grant from the NIGMS (3U24GM132013-02S2) (to K.E.); Meiji Yasuda Life Foundation of Health and Welfare (K.E.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.