Researchers at Queen Mary University of London’s Precision Healthcare University Research Institute (PHURI) and the Berlin Institute of Health (BIH) at Charité – Universitätsmedizin Berlin have identified the genetic causes of Raynaud’s phenomenon (also called Raynaud’s syndrome and Raynaud’s disease). Their findings, published today (October 12) in Nature Communications, could lead to the first effective treatments for people with Raynaud’s.
Understanding Raynaud’s Phenomenon
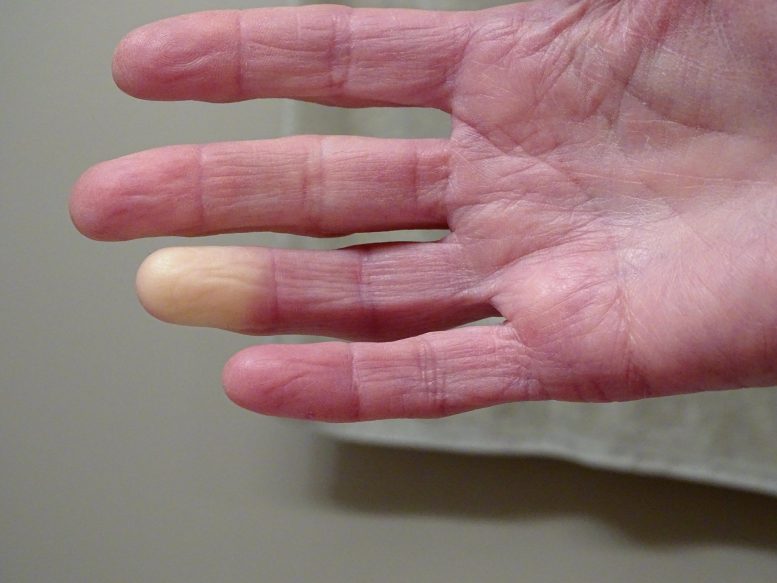
Raynaud’s phenomenon (RP) is a heritable condition that affects blood circulation. It’s a vasospastic condition, which means that small blood vessels near the surface of the skin have spasms that can limit blood flow. People with Raynaud’s often experience pain in their fingers and toes, often alongside changes of color in their skin, due a lack of blood flow during attacks when they’re cold or emotionally stressed. In more serious cases, it can cause severe pain or ulcers.
Around 2-5% of the population is affected by Raynaud’s. Despite it being a common condition, it’s under-investigated and little is understood about the genetic cause of the condition.
Current Treatment and Need for Improved Understanding
There are limited treatments available for RP. Doctors usually advise that the patient use ‘self-management’ strategies such as keeping warm and avoiding triggers of attacks. In severe cases medications can be prescribed, these are ‘repurposed drugs’ usually medicines to lower high blood pressure. These often cause severe side effects in patients. A better understanding of the underlying genetic mechanisms that cause RD is needed to develop safe and effective treatments.
Research Methodology
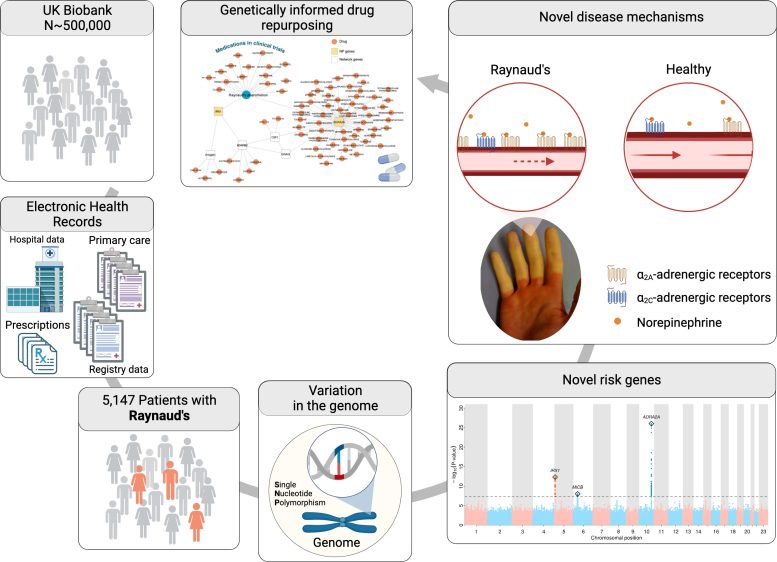
Researchers led by Professor Claudia Langenberg and Professor Maik Pietzner, working across PHURI and the BIH, carried out the largest genetic study of Raynaud’s phenomenon. The team used electronic health records from the UK Biobank, a large-scale biomedical database and research resource containing genetic and health information from half a million UK participants, to identify more than 5,000 people affected by Raynaud’s. The team also used electronic health records from Queen Mary’s Genes & Health study.
The Findings
The researchers discovered variation in two genes that predisposed participants to Raynaud’s phenomenon: One was the alpha-2A-adrenergic receptor for adrenaline, ADRA2A, a classic stress receptor that causes the small blood vessels to contract.
“This makes sense when it’s cold or dangerous, because the body has to supply the inside of the body with blood,” explains Maik Pietzner, Professor of Health Data Modelling at PHURI and group leader at BIH.
“In Raynaud’s patients, this receptor seemed to be particularly active, which could explain the vasospasms, especially in combination with the second gene that we found: This gene is the transcription factor IRX1, which may regulate the ability of blood vessels to dilate.
“If its production is increased, it may activate genes that prevent constricted vessels from relaxing as they would normally do. Together with the overactive adrenaline receptor, this may then lead to the vessels not suppling enough blood for a longer period of time, which leads to the observed white fingers and toes.”
The researchers replicated some of their findings using data from participants of British Bangladeshi and Pakistani origin from Queen Mary’s Genes & Health study.
Potential Implications and Future Research
The researchers’ findings help to understand, for the first time, why the small vessels react so strongly in patients, even apparently without external stimuli, such as exposure to cold.
Dr. Emma Blamont, Head of Research for Scleroderma and Raynaud’s UK (SRUK), said: “Raynaud’s is a painful, chronic condition that affects around one in six people in the UK. We know that attacks can be brought on by certain triggers like cold and stress, but relatively little is known about why some people experience Raynaud’s and others don’t. For the millions of people living with this condition, simple everyday tasks can be a challenge, so research like this, which significantly advances our understanding of Raynaud’s and the role that genetics may play in causing it, is crucial.
“The next step is to confirm these important findings in more diverse population groups and validate the results through functional studies. If successful, these findings could help us unlock more new therapeutic avenues for Raynaud’s leading to better, more targeted, and kinder treatments.”
The findings could lead to recommendations for patients to help manage the condition or its symptoms. For example, the researchers showed that people with a genetic predisposition to low blood sugar levels have an increased risk of Raynaud’s phenomenon, suggesting that patients should possibly avoid longer episodes of low blood sugar.
For Claudia Langenberg, Director of PHURI and Professor of Computational Medicine at BIH, this study exemplifies that integrating genomic and electronic health record data can rapidly help to better understand diseases whose etiology remains unknown. She said:
“Of course, we ultimately hope that our findings will point to novel treatment options. Approved drugs that more or less specifically inhibit the function of ADRA2A, such as the antidepressant mirtazapine, already exist, and our results suggest that these may present alternative treatment options for patients suffering from the symptoms of Raynaud’s.”
Reference: “ADRA2A and IRX1 are putative risk genes for Raynaud’s phenomenon” by Sylvia Hartmann, Summaira Yasmeen, Benjamin M. Jacobs, Spiros Denaxas, Munir Pirmohamed, Eric R. Gamazon, Mark J. Caulfield, Genes & Health Research Team, Harry Hemingway, Maik Pietzner and Claudia Langenberg, 12 October 2023, Nature Communications.
DOI: 10.1038/s41467-023-41876-5
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.