Using diffusion-weighted magnetic resonance, researchers captured pictures of the activation of microglia and astrocytes, two kinds of cells involved in neuroinflammation.
The labs of Dr. Silvia de Santis and Dr. Santiago Canals from the Institute of Neurosciences UMH-CSIC (Alicante, Spain) have used diffusion-weighted magnetic resonance imaging to image brain inflammation not only for the first time but also in great detail.
Data gathering sequences and specialized mathematical models are needed to produce this in-depth “X-ray” of inflammation, which cannot be done with a standard MRI. After developing the technique, the researchers were able to measure the changes in the morphology of the various cell populations contributing to the inflammatory process in the brain.
This significant discovery, which was recently published in the journal Science Advances and may be essential to altering the trajectory of research and treatment of neurodegenerative diseases, was made possible by an innovative strategy created by the researchers.
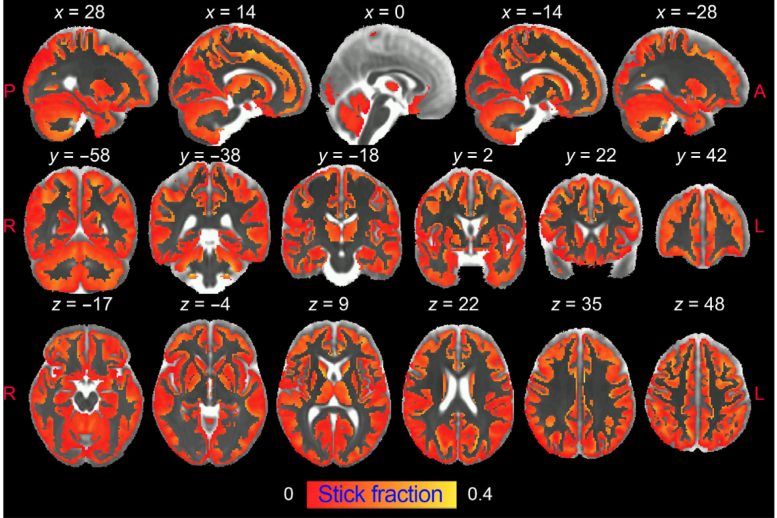
Noninvasive and High-Resolution Inflammation Detection
The study, whose first author is Raquel Garcia-Hernández, shows that diffusion-weighted MRI can noninvasively and differentially detect the activation of microglia and astrocytes, two kinds of brain cells that are at the source of neuroinflammation and its development.
Degenerative brain conditions including Parkinson’s, multiple sclerosis, Alzheimer’s, and other dementias are critical and challenging issues to solve. One of the causes of neurodegeneration and a factor in its progression is chronic inflammation in the brain, which is brought on by the sustained activation of two kinds of brain cells, microglia and astrocytes.
However, there is a lack of non-invasive approaches capable of specifically characterizing brain inflammation in vivo. The current gold standard is positron emission tomography (PET), but it is difficult to generalize and is associated with exposure to ionizing radiation, so its use is limited in vulnerable populations and in longitudinal studies, which require the use of PET repeatedly over a period of years, as is the case in neurodegenerative diseases.
Another drawback of PET is its low spatial resolution, which makes it unsuitable for imaging small structures, with the added drawback that inflammation-specific radiotracers are expressed in multiple cell types (microglia, astrocytes, and endothelium), making it impossible to differentiate between them.
Diffusion-Weighted MRI: A Powerful Alternative
In the face of these drawbacks, diffusion-weighted MRI has the unique ability to image brain microstructure in vivo noninvasively and with high resolution by capturing the random movement of water molecules in the brain parenchyma to generate contrast in MRI images.
In this study, researchers from the UMH-CSIC Neurosciences Institute have developed an innovative strategy that allows imaging of microglial and astrocyte activation in the gray matter of the brain using diffusion-weighted magnetic resonance imaging (dw-MRI).
“This is the first time it has been shown that the signal from this type of MRI (dw-MRI) can detect microglial and astrocyte activation, with specific footprints for each cell population. This strategy we have used reflects the morphological changes validated post-mortem by quantitative immunohistochemistry,” the researchers note.
Differentiating Neuroinflammation and Demyelination
They have also shown that this technique is sensitive and specific for detecting inflammation with and without neurodegeneration so that both conditions can be differentiated. In addition, it makes it possible to discriminate between inflammation and demyelination characteristics of multiple sclerosis.
This work has also been able to demonstrate the translational value of the approach used in a cohort of healthy humans at high resolution, “in which we performed a reproducibility analysis. The significant association with known microglia density patterns in the human brain supports the usefulness of the method for generating reliable glia biomarkers. We believe that characterizing, using this technique, relevant aspects of tissue microstructure during inflammation, noninvasively and longitudinally, can have a tremendous impact on our understanding of the pathophysiology of many brain conditions and can transform current diagnostic practice and treatment monitoring strategies for neurodegenerative diseases,” highlights Silvia de Santis.
Validating the Model with Animal Studies
To validate the model, the researchers have used an established paradigm of inflammation in rats based on intracerebral administration of lipopolysaccharide (LPS). In this paradigm, neuronal viability and morphology are preserved, while inducing, first, activation of microglia (the brain’s immune system cells), and in a delayed manner, an astrocyte response. This temporal sequence of cellular events allows glial responses to be transiently dissociated from neuronal degeneration and the signature of reactive microglia investigated independently of astrogliosis.
To isolate the imprint of astrocyte activation, the researchers repeated the experiment by pretreating the animals with an inhibitor that temporarily ablates about 90% of microglia. Subsequently using an established paradigm of neuronal damage, they tested whether the model was able to unravel neuroinflammatory “footprints” with and without concomitant neurodegeneration. “This is critical to demonstrate the utility of our approach as a platform for the discovery of biomarkers of inflammatory status in neurodegenerative diseases, where both glia activation and neuronal damage are key players,” they clarify.
Finally, the researchers used an established paradigm of demyelination, based on focal administration of lysolecithin, to demonstrate that the biomarkers developed do not reflect the tissue alterations frequently found in brain disorders.
Reference: “Mapping microglia and astrocyte activation in vivo using diffusion MRI” by Raquel Garcia-Hernandez, Antonio Cerdán Cerdá, Alejandro Trouve Carpena, Mark Drakesmith, Kristin Koller, Derek K. Jones, Santiago Canals and Silvia De Santis, 27 May 2022, Science Advances.
DOI: 10.1126/sciadv.abq2923
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.