
Scientists at The Picower Institute for Learning and Memory at MIT have provided comprehensive insight into how serotonin affects behavior in a study using the nematode worm C. elegans, a simple animal model.
The research team identified the functional roles of the worm’s six serotonin receptors by creating 64 different mutant strains, each missing different combinations of receptors. They discovered that three receptors primarily drove the slowing behavior associated with serotonin release, while the other three receptors interacted with the primary receptors and modulated their function. Furthermore, different receptors were found to respond to different patterns of serotonin release. By fluorescently tagging each receptor gene in each neuron across the brain, the team observed how serotonin’s effects worked at a circuit level. The study provides a view of the complexities and opportunities for the development of psychiatric drugs that target the serotonergic system.
Because serotonin is one of the primary chemicals the brain uses to influence mood and behavior, it is also the most common target of psychiatric drugs. To improve those drugs and to invent better ones, scientists need to know much more about how the molecule affects brain cells and circuits both in health and amid disease. In a new study, researchers at The Picower Institute for Learning and Memory at MIT working in a simple animal model present a comprehensive accounting of how serotonin affects behavior from the scale of individual molecules all the way to the animal’s whole brain.
“There have been major challenges in rationally developing psychiatric drugs that target the serotonergic system,” said Steve Flavell, associate professor in The Picower Institute and MIT’s Department of Brain and Cognitive Sciences, and senior author of the study in Cell. “The system is wildly complex. There are many different types of serotonergic neurons with widespread projections throughout the brain and serotonin acts through many different receptors, which are often activated in concert to change the way that neural circuits work.”
These same complexities that scientists face in people are all afoot in the nematode worm C. elegans, but to a more manageably limited degree. C. elegans has only 302 neurons (rather than billions) and only six serotonin receptors (rather than the 14 found in people). Moreover, all C. elegans neurons and their connections have been mapped out and its cells are accessible for genetic manipulation. Finally, Flavell’s team has developed imaging technologies that enable them to track and map neural activity across the worm’s brain simultaneously. For all these reasons, the lab was able to produce a novel study revealing how the far-reaching molecular activity of serotonin changes brain-wide activity and behavior.
“These results provide a global view of how serotonin acts on a diverse set of receptors distributed across a connectome to modulate brain-wide activity and behavior,” the research team wrote in Cell.
The study’s co-lead authors are Picower Institute postdoc Ugur Dag, MIT Brain and Cognitive Sciences graduate student Di Kang, and former research technician Ijeoma Nwabudike, who is now a MD-PhD student at Yale.
Slowing for Savoring
Flavell showed in Cell in 2013 that C. elegans uses serotonin to slow down when it reaches a patch of food and traced its source to a neuron called NSM. In the new study, the team used their many new capabilities developed since then at MIT to examine serotonin’s effects comprehensively.
First, they focused on identifying the functional roles of the worm’s six serotonin receptors. To do that they created 64 different mutant strains covering the different combinations of knocking out the various receptors. For instance, one strain would have just one receptor knocked out while another strain would have all but that one missing and another would be missing three. In each of these worms the team stimulated serotonin release from the NSM neuron to prompt slowing behaviors.
Analysis of all the resulting data revealed at least two key findings: One was that three receptors primarily drove the slowing behavior. The second was that the other three receptors “interacted” with the receptors that drive slowing and modulated how they function. These complex interactions between serotonin receptors in the control of behavior is likely to be directly relevant to psychiatric drugs that target these receptors, Flavell said.
The researchers also gained other important insights into serotonin’s actions. One was that different receptors respond to different patterns of serotonin release in live animals. For example, the SER-4 receptor only responded to sudden increases in serotonin release by the NSM neuron. However, the MOD-1 receptor responded to continuous “tonic” changes in serotonin release by NSM. This suggests that different serotonin receptors are engaged at different times in the live animal.
Brain-Wide Mapping
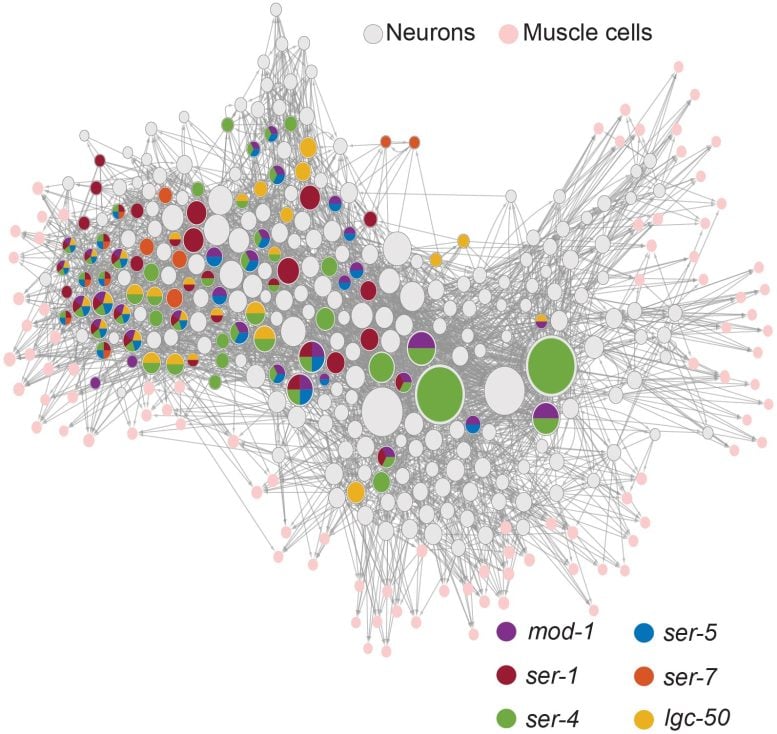
Having teased out the roles of the serotonin receptors in the control of C. elegans behavior, the research team then used their imaging technologies to see how serotonin’s effects worked at a circuit level. For instance, they fluorescently tagged each receptor gene in each neuron across the brain so that they could see all the specific cells that expressed each receptor, providing a brain-wide map of where the serotonin receptors are located in C. elegans. About half of the worm’s neurons express serotonin receptors with some neurons expressing as many as five different types.
Finally, the team used their ability to track all neuron activity (based on their calcium fluctuations) and all behaviors to watch how the serotonergic neuron NSM affected other cells’ activity as worms freely explored their surroundings. About half of the neurons across the worm’s brain changed activity when serotonin was released. Since they knew which exact neurons they were recording from, the research team asked whether knowing which serotonin receptors each cell expressed could predict how they responded to serotonin. Indeed, knowing which receptors were expressed in each neuron and its input neurons gave strong predictive power of how each neuron was impacted by serotonin.
“We performed brain-wide calcium imaging in freely-moving animals with knowledge of cellular identity during serotonin release, providing, for the first time, a view of how serotonin release is associated with changes in activity across the defined cell types of an animal’s brain,” the researchers concluded.
All these findings shed light on the kinds of complexities and opportunities facing drug developers, Flavell noted. The study’s findings show how the effects of targeting one serotonin receptor could depend on how other receptors or the cell types that express them are functioning. In particular, the study highlights how the serotonin receptors act in concert to change the activity states of neural circuits.
Reference: “Dissecting the functional organization of the C. elegans serotonergic system at whole-brain scale” by Ugur Dag, Ijeoma Nwabudike, Di Kang, Matthew A. Gomes, Jungsoo Kim, Adam A. Atanas, Eric Bueno, Cassi Estrem, Sarah Pugliese, Ziyu Wang, Emma Towlson and Steven W. Flavell, 15 May 2023, Cell.
DOI: 10.1016/j.cell.2023.04.023
In addition to Flavell, Dag, Nwabudike and Kang, the paper’s other authors are Matthew Gomes, Jungsoo Kim, Adam Atanas, Eric Bueno, Cassi Estrem, Sarah Pugliese, Ziyu Wang and Emma Towlson.
Study funders included the National Institutes of Health, the National Science Foundation, the McKnight Foundation, the Alfred P. Sloan Foundation, the Picower Institute and the JPB Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.