Researchers develop a method for on-demand hydrogen production, which has potential for use in portable hydrogen fuel cells.
Since the Industrial Revolution, the environmental impacts of energy have posed a concern. Recently, this has driven researchers to search for viable options for clean and renewable energy sources.
Due to its affordability and environmental friendliness, hydrogen is a feasible alternative to fossil fuels for energy applications. However, due to its low density, hydrogen is difficult to transport efficiently, and many on-board hydrogen generation methods are slow and energy intensive.
Researchers from the Chinese Academy of Sciences, Beijing and Tsinghua University, Beijing investigate real-time, on-demand hydrogen generation for use in fuel cells, which are a quiet and clean form of energy. They describe their results in the Journal of Renewable and Sustainable Energy, from AIP Publishing.
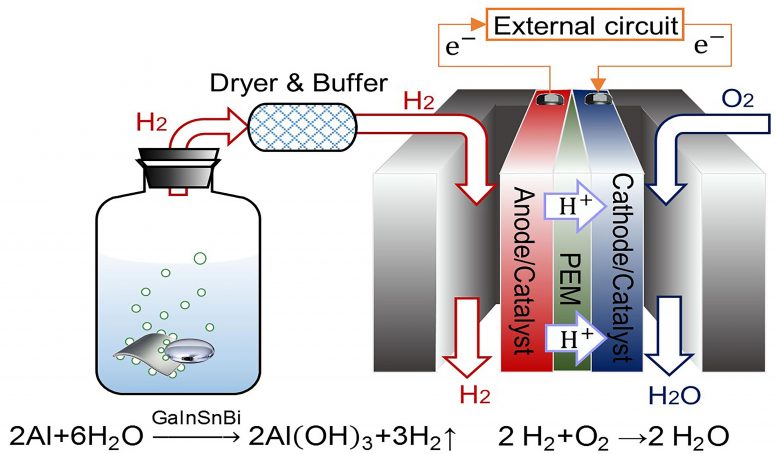
The researchers used an alloy — a combination of metals — of gallium, indium, tin, and bismuth to generate hydrogen. When the alloy meets an aluminum plate immersed in water, hydrogen is produced. This hydrogen is connected to a proton exchange membrane fuel cell, a type of fuel cell where chemical energy is converted into electrical energy.
“Compared with traditional power generation methods, PEMFC inherits a higher conversion efficiency,” said author Jing Liu, a professor at the Chinese Academy of Sciences and Tsinghua University. “It could start rapidly and run quietly. Moreover, a key benefit to this process is that the only product it generates is water, making it environmentally friendly.”
They found the addition of bismuth to the alloy has a large effect on hydrogen generation. Compared to an alloy of gallium, indium, and tin, the alloy including bismuth leads to a more stable and durable hydrogen generation reaction. However, it is important to be able to recycle the alloy in order to further reduce cost and environmental impact.
“There are various problems in existing methods for post-reaction mixture separation,” Liu said. “An acid or alkaline solution can dissolve aluminum hydroxide but also causes corrosion and pollution problems.”
Other byproduct removal methods are difficult and inefficient, and the problem of heat dissipation in the hydrogen reaction process also needs to be optimized. Once these difficulties are resolved, this technology can be used for applications from transportation to portable devices.
“The merit of this method is that it could realize real-time and on-demand hydrogen production,” said Liu. “It may offer a possibility for a green and sustainable energy era.”
Reference: “Instant hydrogen production using Ga-In-Sn-Bi alloy-activated Al-water reaction for hydrogen fuel cells” by Shuo Xu, Yuntao Cui, Lixiang Yang and Jing Liu, 28 January 2020, Journal of Renewable and Sustainable Energy.
DOI: 10.1063/1.5124371
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
7 Comments
It’s useless & too expensive! The best is potassium hydroxide.
Neat stuff… We’re gettin’ there!
Water vapour is a greenhouse gas. Fuel cells produce water vapour!
I’m no scientist…but doesn’t water vapor generally end up in clouds, and thence rain? That was the Water Cycle I learned about when I was around 5 or so.
Tom. How useful that would be in parts of the African continent, and Australia.Hydrogen created by the sun, turned into combustible gas and water for crops? Wow!
I have been a fan of hydrogen for the past week years. In 2008 BMW showcased a BMW that had an internal combustion engine. This engine would also run on gasoline. What an amazing bridge to get us there. The waste output was water. Just like these fuel cells. Whatever happened to that technology?
Utter bullsnot
Free energy always comes from free previous processes that are not accounted for.
So 2Al+6H2O => 2Al(OH)3 + 3H2.
This isn’t remotely clean hydrogen, it says so right in the eqn, Al goes in and turns in to Al-OH, ie slush waste that has to be recycled very similar to bauxite.
The Al is the energy carrier, during the process I expect half the energy embedded in the Al turns into heat and the remainder into the hydrogen as chemical energy. Then the fuel cell, maybe another half is turned into more heat and electricity. Overall efficiency is between 25-40% eff as in J of chemical energy in & out. But wait there is so much more.
To make the Al requires copious electrical energy to split the Al bonds with O2 in the bauxites mined material or this recycled slush.
If we start with the bauxite and electrical energy in Je we are using electrical energy with about 3x the chemical energy value of the chemical energy that made it.
So now we go back further, coal => electricity => Al(O..) => Al then Al… => H2 then H2 => electricity and we could have had the electrity directly from coal and skipped the issue of passing through Al then Al =>H2 and the fuel cell. Skipping these extra steps likely means indirect electricty from coal is maybe 10 times worse than directly burning coal.
Utter green washing, a complicated chain of energy losses.
The only useful thing here is that the metal as energy carrier is a compact battery that might be useful in airplanes, but it would be far better to directly make JP14 with nuclear heat. Or just put a coal burner on the plane as if it were a steam train.
I am not even a chemist, but this is just basic chemistry and physics.