
Lakes Could Be Suitable for the Extraction of Methane for Energy Production
Lakes store huge amounts of methane. In a new study, environmental scientists at the University of Basel offer suggestions for how it can be extracted and used as an energy source in the form of methanol.
Discussion about the current climate crisis usually focuses on carbon dioxide (CO2). The greenhouse gas methane is less well known, but although it is much rarer in the atmosphere, its global warming potential is 80 to 100 times greater per unit.
More than half the methane caused by human activities comes from oil production and agricultural fertilizers. But the gas is also created by the natural decomposition of biomass by microbes, for example in lakes. In their most recent publication, Maciej Bartosiewicz, postdoc in the Department of Environmental Sciences at the University of Basel, and Professor Moritz Lehmann, head of the biogeochemistry research group, outline the potential and theoretical possibilities for using methane from lakes and other freshwater bodies for sustainable energy production.
Methane from lakes and water reservoirs makes up about 20% of global natural methane emissions. “That would theoretically be enough to meet the world’s energy needs,” says Bartosiewicz. Lakes continuously absorb CO2 from the atmosphere through the growth of phytoplankton. Microbes convert the carbon, fixed by photosynthesis, into methane when they process biomass. That way, carbon bound in the methane remains within the natural cycle during combustion. Fossil fuels could be partially replaced by “natural” renewable methane. Methane gas is already burned in gas-fired power plants for electricity production and used as a fuel in the form of liquid methanol.
Swiss Lakes As Huge Energy Stores
The idea described in the article isn’t completely new: since 2016, methane in Lake Kivu between Rwanda and the Democratic Republic of Congo has been extracted from a depth of 260 meters (850 feet), cleaned, and used for energy supply directly via generators. “Methane occurs in high concentrations in large quantities on the lake bed there,” explains Bartosiewicz. “The methane concentration is about 100 times higher than in ordinary lakes.” Low concentrations made extracting methane from conventional lakes seem too technically difficult until a few years ago. But new microporous membranes made of polymeric materials now allow the gas to be separated from the water much more efficiently.
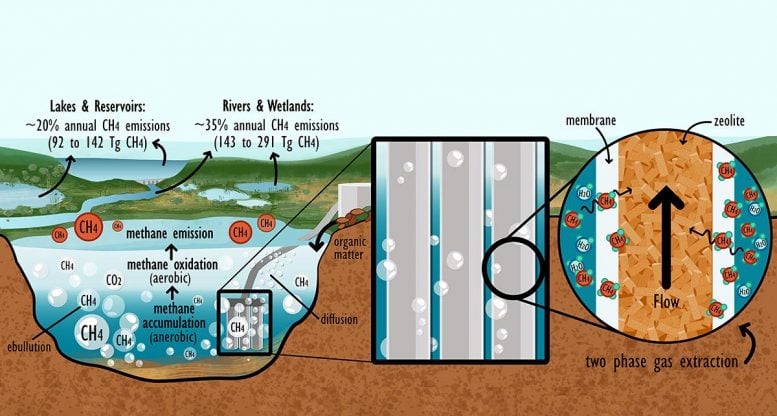
The researchers have made the first concrete proposals in this regard: using a hydrophobic gas-liquid membrane contactor (GLMC), a methane-containing gas mixture can be separated from water and the methane concentrated. Zeolite minerals are particularly suitable for enrichment, since hydrophobic crystalline substances can adsorb and release gases.
“Switzerland is practically made for methane extraction from lakes,” says Bartosiewicz. The reason is the country’s large number of reservoirs: “We have recently learned that they also produce and emit into the atmosphere large quantities of methane.” In addition, the chair of heterogeneous catalysis at ETH Zurich, which also participated in the study, has extensive expertise in adsorption processes using zeolites.
Potential Positive Effects on Ecosystems
“With our idea, we wanted to start a broad discussion about the potential, feasibility, and risks of a technology like this,” says Bartosiewicz. “Until now, no studies have addressed the effects of methane removal on lake ecosystem functioning, but no immediate negative effects can be foreseen with our current understanding.” However, removing excess carbon could even help curb excessive phytoplankton bloom formation and reduce natural greenhouse gas emissions from lakes. More work is needed before any practical implementation of this initial theoretical idea, says Bartosiewicz. But he’s convinced: “This concept could one day make an important contribution to reaching our climate goals.”
Reference: “Tapping Freshwaters for Methane and Energy” by Maciej Bartosiewicz, Przemyslaw Rzepka and Moritz F. Lehmann, 5 March 2021, Environmental Science & Technology.
DOI: 10.1021/acs.est.0c06210
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.