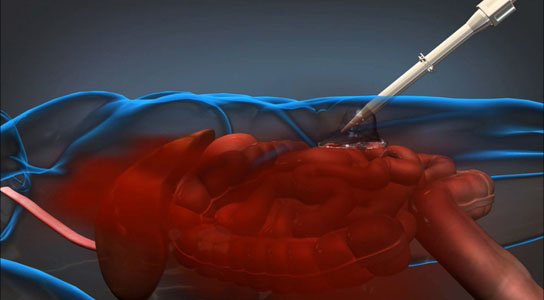
The foam-based product developed by Arsenal Medical for DARPA can be injected into a wounded servicemember on the battlefield to slow blood loss until the patient can be transported to an appropriate medical facility. Credit: DARPA
Future combat medics could save the lives of soldiers, preventing them from bleeding out on battlefields, by injecting medical foam into their bodies.
The scientists presented their findings at the 2012 Annual Meeting of the American Association for the Surgery of Trauma in Kauai, Hawaii. Currently, there is little that can be done to stem internal bleeding before patients reach necessary treatment facilities. The resulting blood loss often leads to death from what would otherwise be potentially survivable wounds.
As DARPA’s Wound Stasis System program evolved, a better possible solution emerged. Wound Stasis performer Arsenal Medical, Inc. developed a foam-based product that can control hemorrhaging in a patient’s intact abdominal cavity for at least one hour, based on swine injury model data. The foam is designed to be administered in battlefield situations by combat medics and can easily be removed by doctors at medical facilities.
Pre-clinical data demonstrated the ability of the foam to treat severe hemorrhage for up to three hours in a model of lethal liver injury. During testing, minimally invasive application of the product reduced blood loss six-fold and increased the rate of survival at three hours post-injury to 72 percent from the eight percent observed in controls.
The foam is actually a polyurethane polymer that forms inside a patient’s body upon injection of two liquid phases, a polyol phase and an isocyanate phase, into the abdominal cavity. As the liquids mix, two reactions are triggered. First, the mixed liquid expands to approximately 30 times its original volume while conforming to the surfaces of injured tissue. Second, the liquid transforms into solid foam capable of providing resistance to intra-abdominal blood loss. The foam can expand through pooled and clotted blood despite the significant hydrostatic force of an active hemorrhage.
In tests, removal of the foam took less than one minute following incision by a surgeon. The foam was removed by hand in a single block, with only minimal amounts remaining in the abdominal cavity, and with no significant adherence of tissue to the foam. Features appearing in relief on the extracted foam showed conformal contact with abdominal tissues and partial encapsulation of the small and large bowels, spleen, and liver. Blood absorption was limited to near the surface of the foam; the inside of the foam block remained almost uniformly free of blood.
The foam technology could affect up to 50 percent of potentially survivable battlefield wounds. DARPA awarded a $15.5 million Phase II contract to Arsenal Medical to continue development of the treatment system and support regulatory submission. DARPA anticipates continuing the Wound Stasis program through at least FDA approval of a prototype device.









Be the first to comment on "Foam Injections Could Prevent Deaths on the Battlefield"