
Researchers demonstrate that organoids react to external sensory stimuli using cutting-edge recording technology.
A group of engineers and neuroscientists have shown, for the first time, that brain organoids implanted in mice form functional connections to the mice’s cortex and respond to external sensory stimuli. The team observed the organoids reacting to visual stimuli similarly to the surrounding tissues, thanks to a transparent graphene microelectrode array and two-photon imaging system that allowed real-time monitoring over several months.
The study, which was recently published in the journal Nature Communications, was led by Duygu Kuzum, a researcher in the Electrical and Computer Engineering Department at UC San Diego. Collaborators include researchers from Anna Devor’s lab at Boston University, Alysson R. Muotri’s lab at UC San Diego, and Fred H. Gage’s lab at the Salk Institute.

Human cortical organoids are derived from human induced pluripotent stem cells, which are usually derived themselves from skin cells. These brain organoids have recently emerged as promising models to study the development of the human brain, as well as a range of neurological conditions.
But until now, no research team had been able to demonstrate that human brain organoids implanted in the mouse cortex were able to share the same functional properties and react to stimuli in the same way. This is because the technologies used to record brain function are limited, and are generally unable to record activity that lasts just a few milliseconds.
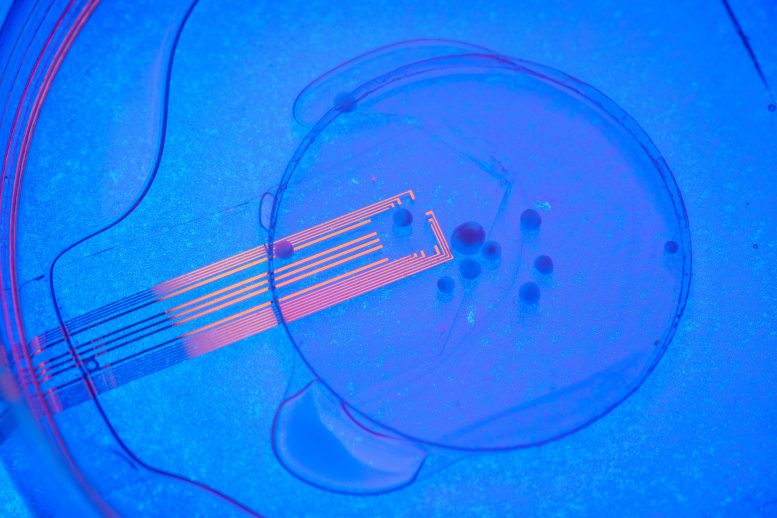
The UC San Diego-led team was able to solve this problem by developing experiments that combine microelectrode arrays made from transparent graphene, and two-photon imaging, a microscopy technique that can image living tissue up to one millimeter in thickness.
Observing Organoid Responses to Sensory Stimuli
“No other study has been able to record optically and electrically at the same time,” said Madison Wilson, the paper’s first author and a Ph.D. student in Kuzum’s research group at UC San Diego. “Our experiments reveal that visual stimuli evoke electrophysiological responses in the organoids, matching the responses from the surrounding cortex.”
The researchers hope that this combination of innovative neural recording technologies to study organoids will serve as a unique platform to comprehensively evaluate organoids as models for brain development and disease, and investigate their use as neural prosthetics to restore function to lost, degenerated, or damaged brain regions.
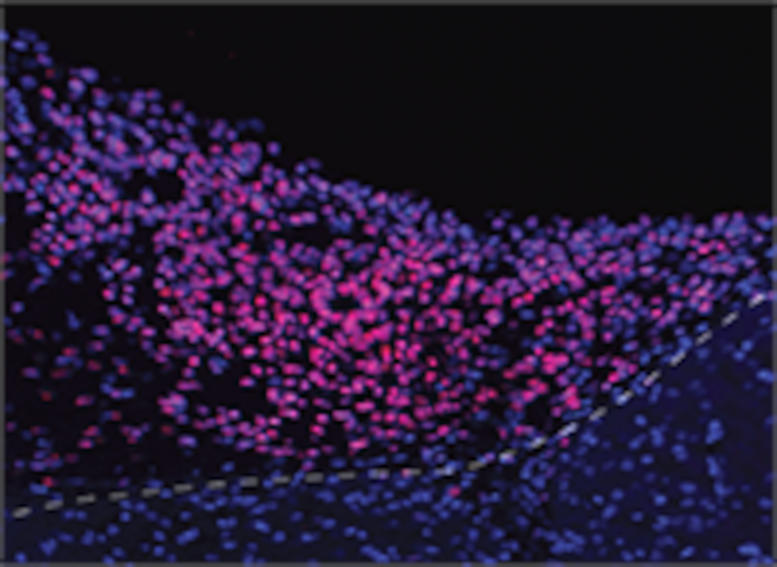
“This experimental setup opens up unprecedented opportunities for investigations of human neural network-level dysfunctions underlying developmental brain diseases,” said Kuzum.
Kuzum’s lab first developed the transparent graphene electrodes in 2014 and has been advancing the technology since then. The researchers used platinum nanoparticles to lower the impedance of graphene electrodes by 100 times while keeping them transparent. The low-impedance graphene electrodes are able to record and image neuronal activity at both the macroscale and single cell levels.
By placing an array of these electrodes on top of the transplanted organoids, researchers were able to record neural activity electrically from both the implanted organoid and the surrounding host cortex in real-time. Using two-photon imaging, they also observed that mouse blood vessels grew into the organoid providing necessary nutrients and oxygen to the implant.
Neural Activity and Visual Stimulus Responses
Researchers applied a visual stimulus–an optical white light LED–to the mice with implanted organoids, while the mice were under two-photon microscopy. They observed electrical activity in the electrode channels above the organoids showing that the organoids were reacting to the stimulus in the same way as the surrounding tissue. The electrical activity propagated from the area closest to the visual cortex in the implanted organoids area through functional connections.
In addition, their low-noise transparent graphene electrode technology enabled the electrical recording of spiking activity from the organoid and the surrounding mouse cortex. Graphene recordings showed increases in the power of gamma oscillations and phase locking of spikes from organoids to slow oscillations from mouse visual cortex. These findings suggest that the organoids had established synaptic connections with surrounding cortex tissue three weeks after implantation, and received functional input from the mouse brain. Researchers continued these chronic multimodal experiments for eleven weeks and showed functional and morphological integration of implanted human brain organoids with the host mice’s cortex.
Future Applications and Disease Modeling
The next steps include longer experiments involving neurological disease models, as well as incorporating calcium imaging in the experimental setup to visualize spiking activity in organoid neurons. Other methods could also be used to trace axonal projections between organoid and mouse cortex.
“We envision that further along the road, this combination of stem cells and neurorecording technologies will be used for modeling disease under physiological conditions; examining candidate treatments on patient-specific organoids; and evaluating organoids’ potential to restore specific lost, degenerated, or damaged brain regions,” Kuzum said.
Reference: “Multimodal monitoring of human cortical organoids implanted in mice reveal functional connection with visual cortex” by Madison N. Wilson, Martin Thunemann, Xin Liu, Yichen Lu, Francesca Puppo, Jason W. Adams, Jeong-Hoon Kim, Mehrdad Ramezani, Donald P. Pizzo, Srdjan Djurovic, Ole A. Andreassen, Abed AlFatah Mansour, Fred H. Gage, Alysson R. Muotri, Anna Devor and Duygu Kuzum, 26 December 2022, Nature Communications.
DOI: 10.1038/s41467-022-35536-3
The study was funded by the National Institutes of Health and the Research Council of Norway, as well as the National Science Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.