A revolutionary genetic score fuses rare and common gene data to predict, and potentially prevent, deadly heart rhythm disorders.
- Researchers used whole genome sequencing to unite three major approaches to genetic testing—monogenic, polygenic, and full-genome analysis—creating a powerful new way to predict heart rhythm risks.
- Experts say more doctors should embrace genetic testing, though many still lack the training and tools to use it effectively in patient care.
- The discovery opens the door to highly personalized, targeted treatments tailored to each person’s unique genetic makeup.
Breakthrough in Predicting Irregular Heartbeats
In a new study from Northwestern Medicine, researchers have created a more accurate genetic risk score that helps determine whether someone is likely to develop arrhythmia, a condition that causes an irregular heartbeat and can lead to serious health issues such as atrial fibrillation (AFib) or sudden cardiac death.
This new method not only enhances how heart disease risk is predicted but also establishes a broad framework for genetic testing that, according to the research team, could eventually be used to assess many other complex, genetically influenced diseases, including cancer, Parkinson’s Disease, and autism.
A “Roadmap” for Genetic Testing Innovation
“It’s a very cool approach in which we are combining rare gene variants with common gene variants and then adding in non-coding genome information. To our knowledge, no one has used this comprehensive approach before, so it’s really a roadmap of how to do that,” said co-corresponding author Dr. Elizabeth McNally, director of the Center for Genetic Testing and a professor of medicine in the division of cardiology and of biochemistry and molecular genetics at Northwestern University Feinberg School of Medicine.
According to the authors, the findings pave the way for precision therapies that can be customized to an individual’s complete genetic makeup. This advancement could also allow physicians to identify patients at risk long before any symptoms appear.
The research, which included 1,119 participants, is being published today (November 11) in Cell Reports Medicine.
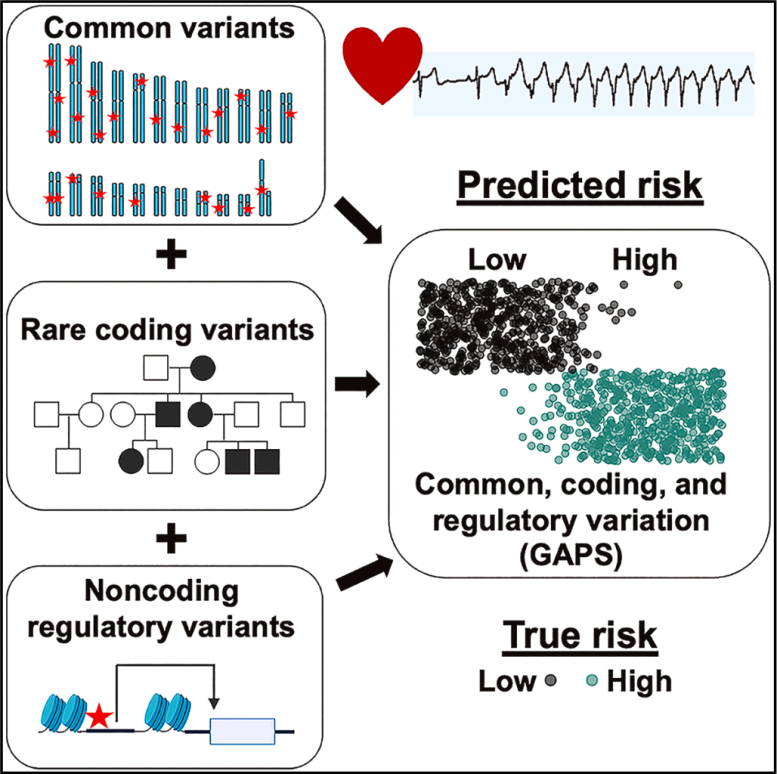
Combining All Three Types of Genetic Testing
At present, genetic testing typically relies on one of three separate methods:
- Monogenic testing: Detects rare mutations in a single gene, similar to finding a typo in one word.
- Polygenic testing: Examines multiple common gene variants to gauge overall risk, much like analyzing the tone of a chapter.
- Genome sequencing: Reads the entire genetic code, comparable to reading a book from start to finish.
Bridging the Genetic Testing Silos
“Genetic researchers, companies, and geneticists often operate in silos,” McNally said. “The companies that offer gene panel testing are not the same ones that provide polygenic risk scores.”
In this project, the Northwestern team combined information from all three types of genetic data to build a complete, 360-degree view of disease risk. This integrated approach makes it possible to detect rare mutations, measure cumulative genetic effects, and identify subtle patterns across the full genome.
“When you sequence the whole genome, you can say, ‘Let me look at this cardiomyopathy gene component, the gene panel, and the polygenic component.’ By combining the data together, you get a very high odds ratio of identifying who is at highest risk, and that’s where we think this approach can really improve upon what is currently used,” McNally said.
Why More Physicians Should Order Genetic Testing
Traditionally, cardiologists assess heart risk by evaluating symptoms, family history, and diagnostic tests like EKGs, echocardiograms, and MRIs. McNally also incorporates genetic testing into her practice, she said.
“It helps me manage that patient better, know who’s at greatest risk, and if we think the risk is really high, we’ll recommend defibrillators for patients like that,” McNally said. “Knowledge is power.”
The Challenge of Low Testing Uptake
Despite its potential, genetic testing remains underutilized, McNally said, adding that it’s estimated that only 1 to 5% of people who should receive genetic testing actually do. Even in cancer care, where genetic links are well established, only 10 to 20% of patients undergo testing.
“We need to improve uptake,” McNally said. “The biggest challenge is a workforce that isn’t trained in how to use genetic testing. As polygenic risk scores become more common, our approach will become even more valuable.”
Inside the Study: How Researchers Did It
The scientists recruited 523 participants who had arrhythmias, and some of these people also had heart failure. The team meticulously went through every case, examining every record to ensure the participants really had had arrhythmias, including looking at data directly from patients’ devices. Finally, they sequenced the patients’ genomes, using monogenetic and polygenetic testing, to determine a risk score and compared the results to the genomes of 596 control participants from the NUgene biobank aged 40 and up with no known cardiac disease history.
“It was painstaking going through 500-plus records and making sure that the people in the study really belonged in the study,” McNally said.
Reference: “A combined genomic arrhythmia propensity score delineates cumulative risk” by Tanner O. Monroe, Megan J. Puckelwartz, Lorenzo L. Pesce, Samuel D. Kearns, Patrick Page, Nora Ibrahim, Zachary T. Weber, Emmanuel I. Ugwor, Marcelo A. Nóbrega, Prince Kannankeril, Laura J. Rasmussen-Torvik, Lisa M. Dellefave-Castillo, Alfred L. George, Gregory Webster and Elizabeth M. McNally, 11 November 2025, Cell Reports Medicine.
DOI: 10.1016/j.xcrm.2025.102455
Other Northwestern study authors include Tanner Monroe, Megan Puckelwartz, Lorenzo Pesce, Dr. Alfred George and Dr. Gregory Webster.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.