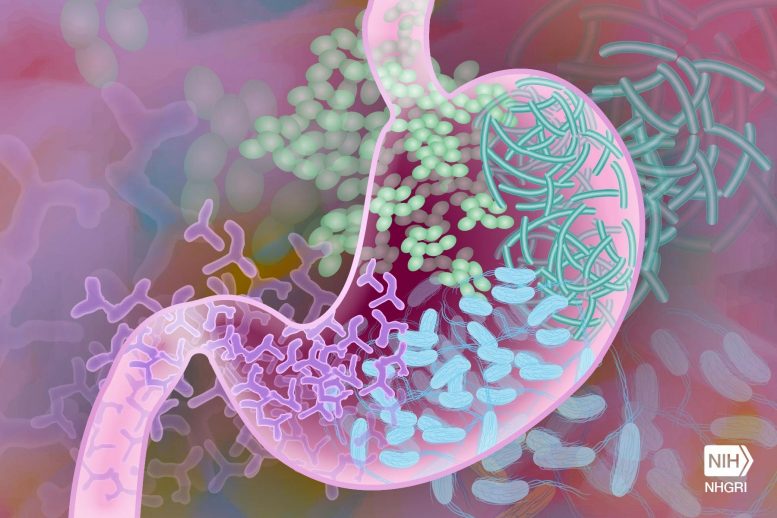
Essential nutrient passes between cells via ‘jumping genes.’
Your gut bacteria need vitamin B12 just as much as you do. Though DNA is usually passed from parent to child, new research shows gut bacteria transfer genes through “sex” in order to take their vitamins.
The majority of living cells cannot function without vitamin B12. As a consequence, it is fiercely competed for in nature. According to recent UC Riverside research, beneficial gut microbes may share this valuable resource with one another via a process known as bacterial sex.
“The process involves one cell forming a tube that DNA can pass through to another cell,” said UCR microbiologist and study lead Patrick Degnan. “It’s as if two humans had sex, and now they both have red hair.”
For many years, scientists have been aware of this phenomenon and its capacity to transmit so-called “jumping genes” across different species. Up until now, the majority of researched instances have been responsible for assisting bacterial cells to survive when individuals consume antibiotics.
“We’re excited about this study because it shows that this process isn’t only for antibiotic resistance. The horizontal gene exchange among microbes is likely used for anything that increases their ability to survive, including sharing vitamin B12,” Degnan said.
Results of the study have been published in the journal Cell Reports.
Previously, Degnan worked on a project in which he and his colleagues identified an important transporter responsible for getting B12 into gut microbial cells. More recently, he was studying jumping genes, trying to identify what kinds of information they were transferring. Quickly, Degnan recognized the vitamin B12 transporters as the cargo.
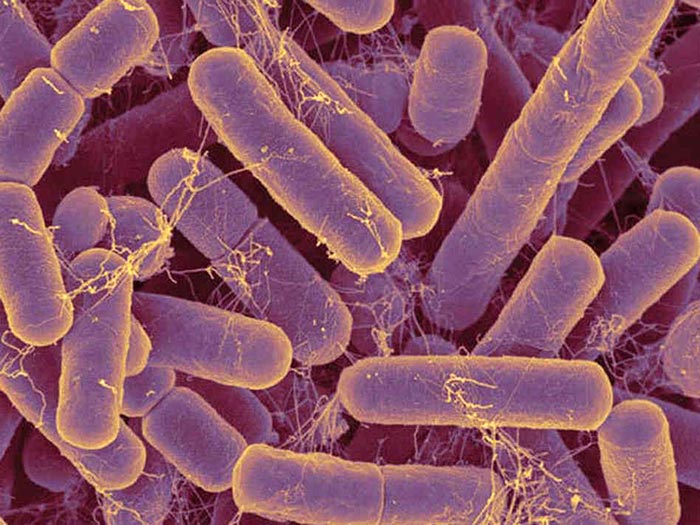
Degnan and his colleagues combined bacteria that could carry B12 and others that couldn’t in order to verify their suspicions. The bacteria had the chance to create a sex pilus, a tube that aided in the transfer, because they were on a plate together. They discovered that bacteria that had previously been unable to transport B12 were all still alive and had gained the genes necessary to do so.
They did a second experiment examining the entire genome of the bacteria.
“In a given organism, we can see bands of DNA that are like fingerprints. The recipients of the B12 transporters had an extra band showing the new DNA they got from a donor,” Degnan said.
Not only was the experiment successful in test tubes, but also inside mice.
The type of beneficial gut bacteria used in the study are Bacteroides, which reside in the large intestines of most people. One of their most important services to humans is breaking down complex carbohydrates for energy.
“The big, long molecules from sweet potatoes, beans, whole grains, and vegetables would pass through our bodies entirely without these bacteria. They break those down so we can get energy from them,” Degnan explained.
Bacteroides, along with other bacteria, also give our guts a barrier layer that can help restrict pathogens from invading. For example, previous research led by co-author Ansel Hsiao, also at UC Riverside, shows some humans have communities of microbes in their gut that make them more resistant to cholera.
Learning how to keep these bacteria healthy could also help benefit people, given the important services they perform.
“There’s no one way to have a healthy microbiome, but generally, having a diverse community of anaerobic bacteria is a healthy thing and can have beneficial effects,” Degnan said.
Reference: “Mobilization of vitamin B12 transporters alters competitive dynamics in a human gut microbe” by Katie A. Frye, Varadh Piamthai, Ansel Hsiao and Patrick H. Degnan, 28 December 2021, Cell Reports.
DOI: 10.1016/j.celrep.2021.110164
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Worst contents or misleading information. Pics are quite opposite and wrong with the content