Researchers at Oregon State University and Oregon Health & Science University are working on a treatment that holds great promise for improving the lives of cystic fibrosis patients.
Cystic fibrosis is a progressive genetic disorder that results in persistent lung infection and afflicts 30,000 people in the U.S., with about 1,000 new cases diagnosed every year.
More than three-quarters of patients are diagnosed by age 2, and despite steady advances in alleviating complications, the median life expectancy of CF patients is still just 40 years.
One faulty gene – the cystic fibrosis transmembrane conductance regulator, or CFTR – causes the disease, which is characterized by lung dehydration and mucous buildup that blocks the airway.
To date more than 300 different disease-conferring mutations have been found, meaning a lot of variance from patient to patient in how the disease progresses.
Treatment to “rescue” the mutant protein produced by the bad gene is available to some patients, although almost one patient in five discontinues therapy because of the high cost and the side effects.
Also, about 30 percent of patients have a gene mutation with no protein rescue treatment option, and toxicity concerns limit protein rescue therapy to patients older than age 6.
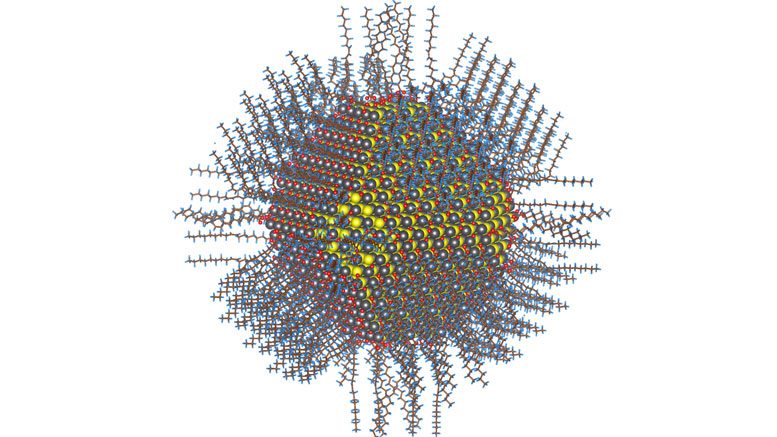
Scientists and clinicians at OSU and OHSU have demonstrated proof-of-concept for a much better means of therapy: loading chemically modified CFTR messenger RNA into lipid-based nanoparticles, creating molecular medicine that could simply be inhaled at home.
Rather than trying to mend the patients’ existing mutant protein behind the mucous and lung dryness, the mRNA-loaded nanoparticle approach causes the cells to actually make the correct protein – that allows cells to properly regulate chloride and water transport, which is critical to healthy respiratory function.
Findings were published in Molecular Therapy.
“It’s a platform technology for correcting monogenic disorders and allows the same therapy to be effective for treating all cystic fibrosis patients,” said corresponding author Gaurav Sahay, assistant professor of pharmaceutical sciences at the OSU/OHSU College of Pharmacy.
Other types of treatments had to be patient-specific because of all of the different mutations, but this therapy works independent of patient genotype.
“And these systems can be repeatedly administered to a patient and the effects are reversible if someone needs to stop the therapy for any reason,” Sahay said.
Sahay and fellow researchers including Kelvin MacDonald, an OHSU physician who treats cystic fibrosis patients, successfully tested the mRNA-loaded nanoparticles in culture with patient-derived bronchial epithelial cells and also in mice that had had their CFTR gene knocked out.
“We made a one-size-fits-all treatment,” said the study’s first author, Ema Robinson, a faculty research assistant in the College of Pharmacy. “We’re adding back the gene that makes the protein so the airways can be rehydrated, and we can adjust the dosage as the disease gets better.”
Robinson notes that a big problem with existing cystic fibrosis therapy is patient compliance.
“If you have a kid you have to bring to the hospital six hours a week on three different days, that could become logistically difficult and you might not always be able to make it happen,” she said. “And the drugs often make them quite sick. We hope that repeated doses of nanoparticles would not make people sick, and would also let patients stay at home and likely increase compliance.”
Joining Robinson, Sahay, and MacDonald on the study were OSU colleagues Kai Slaughter, Madison McKinney, Siddharth Patel, and Conroy Sun.
Supporting this research were the Cystic Fibrosis Foundation, the National Institute of Biomedical Imaging and Bioengineering, the National Institute of General Medical Sciences, and the OSU/OHSU College of Pharmacy.
Reference: “Autologous CD19-Targeted CAR T Cells in Patients with Residual CLL following Initial Purine Analog-Based Therapy” Molecular Therapy.
DOI: 10.1016/j.ymthe.2018.05.018
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.