Two Phase 3, randomized, placebo-controlled, double-blind clinical trials testing whether experimental monoclonal antibodies (mAbs) can prevent infection by SARS-CoV-2 coronavirus are now enrolling healthy adults at clinical trial sites in the United States. Many of the trial sites and study investigators are part of the COVID-19 Prevention Network (CoVPN), recently established by the National Institute of Allergy and Infectious Diseases (NIAID), one of the National Institutes of Health. SARS-CoV-2 is the virus that causes coronavirus disease 2019 (COVID-19). The trials are enrolling adults who are at risk of infection due to close contact at work or home to persons with SARS-CoV-2 infection.
“The COVID-19 Prevention Network is designed to conduct large-scale trials rapidly and efficiently,” said NIAID Director Anthony S. Fauci, M.D. “This network will allow us to test the safety and efficacy of monoclonal antibodies and other preventive measures to help identify how best to reduce the level of SARS-CoV-2 infection and ultimately end the COVID-19 pandemic.”
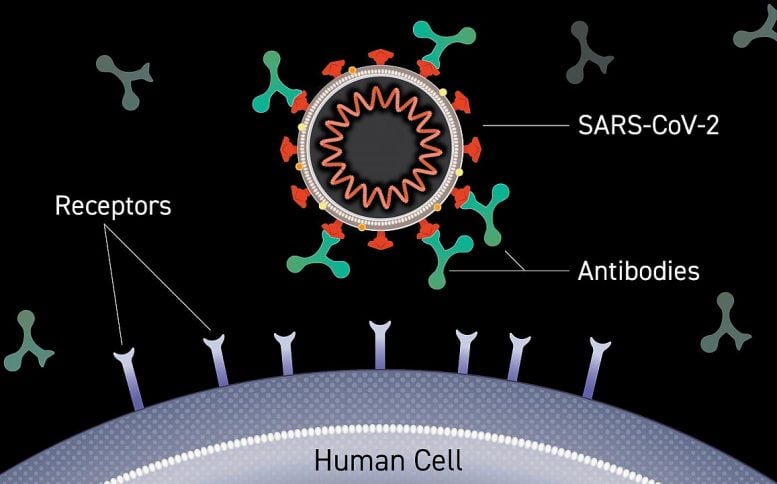
Monoclonal antibodies are laboratory-made versions of proteins naturally produced by the immune system in response to invading viruses or other pathogens. Neutralizing antibodies, whether natural or monoclonal, can bind directly to portions of viruses that they use to attach to and enter cells, preventing them from initiating the infection cycle. Monoclonal antibodies may provide short-term protection from SARS-CoV-2 and could serve as important components of the COVID-19 pandemic response until vaccines become available.
One trial is being conducted jointly by NIAID and trial sponsor Regeneron Pharmaceuticals of Tarrytown, New York. It will evaluate Regeneron’s investigational double mAb combination, REGN-COV-2, which is designed to bind to two points on the SARS-CoV-2 spike protein and prevent it from entering healthy cells. The trial will enroll approximately 2,000 asymptomatic adults who are household contacts of persons with SARS-CoV-2 infection. Participants must have been in close contact (typically due to residing at the same address) with the infected person in a 96-hour window preceding administration of either REGN-CoV-2 or placebo. In addition to assessing safety, the trial will seek to define whether REGN-COV-2 can prevent infection or disease symptoms in those already infected. The efficacy assessment will be a one-month period following administration of REGN-COV-2 or placebo. All trial participants will be followed for safety for seven months after efficacy assessment period ends.
Additional details about this trial are available at clinicaltrials.gov using the identifier NCT04452318. Interested participants can also visit the CoVPN website for details. Doctors or potential participants may also contact the sponsor’s Clinical Trials Administrator at 844-734-6643 or [email protected] for information on enrolling.
A second trial, sponsored by Eli Lilly and Company of Indianapolis, Indiana, and implemented in collaboration with NIAID, will evaluate LY-CoV555, a mAb isolated from a recovered COVID-19 patient by scientists at AbCellera (Vancouver, British Columbia, Canada) and the NIAID Vaccine Research Center, and developed by Eli Lilly and Company. This trial will assess whether LY-CoV555 can prevent SARS-CoV-2 infection among people at high risk of exposure due to residing or working in skilled nursing or assisted living facilities. Within one week of identification of a case of SARS-CoV-2 infection at a facility, study investigators will enroll trial volunteers and evaluate the prevention efficacy and safety of LY-CoV555, compared to placebo, over an 8-week period. The trial will also evaluate efficacy in preventing symptoms of a given severity in those already infected. Participants will continue to be followed for safety for an additional 16 weeks. Up to 2,400 participants will be randomized to receive intravenous infusion of either LY-CoV555 or placebo.
Additional information about this trial is available at clinicaltrials.gov using the identifier NCT04497987. Clinical investigators, hospitals or clinical sites interested in participating in one of Lilly’s clinical trials for a potential COVID-19 treatment, should call 1-877-CT-LILLY (1-877-285-4559) or email [email protected].
NIAID conducts and supports research—at NIH, throughout the United States, and worldwide—to study the causes of infectious and immune-mediated diseases, and to develop better means of preventing, diagnosing and treating these illnesses.
About the National Institutes of Health (NIH), the nation’s medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Interesting that there are studies in many countries of antibodies being produced in a variety of animals – horses, llamas and mention of these efforts in any of the literature on antibody treatments