Researchers have identified a natural compound from pomegranate leaves and branches that can directly dismantle the protein aggregates responsible for transthyretin amyloidosis.
Pomegranate trees are best known for their fruit, but researchers at Kumamoto University are pointing to a different part of the plant as a potential source of new medicine. The team reports that a compound found in pomegranate leaves and branches can directly break apart the protein clumps involved in transthyretin (TTR) amyloidosis, a progressive disorder that can seriously damage the nerves and the heart.
In iScience, the researchers describe 1,2,3,4,6-penta-O-galloyl-β-D-glucose (PGG) as an “amyloid disruptor.” In TTR amyloidosis, transthyretin, a transport protein, can lose its normal shape and stack into tough, insoluble amyloid fibrils that lodge in organs. Many existing approaches focus on keeping the protein stable or lowering how much of it is made. The problem is that once deposits are established, slowing new buildup does not necessarily remove what is already there.
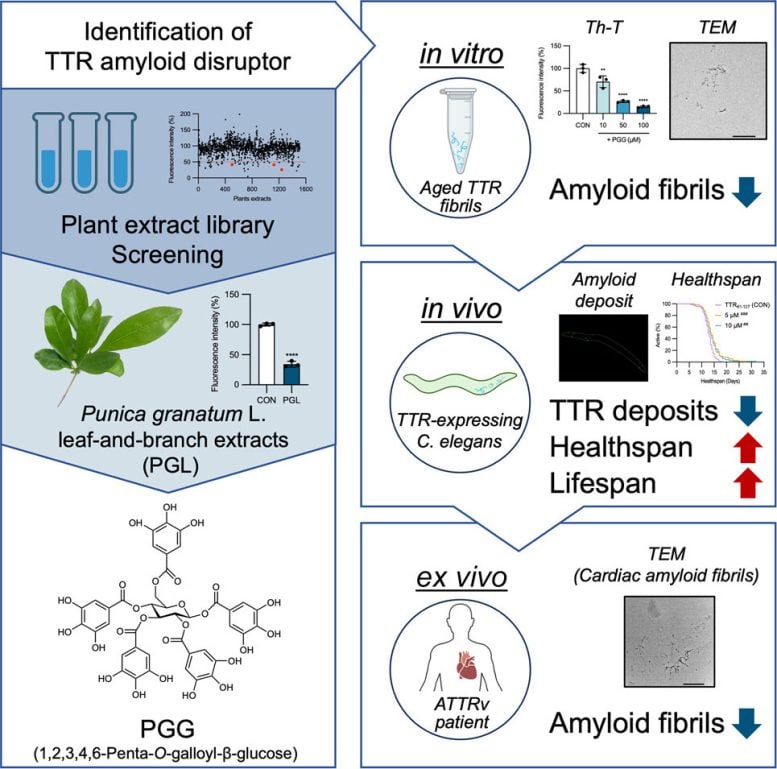
To address this unmet need, the group screened 1,509 plant extracts from a natural product library. Extracts from pomegranate (Punica granatum) leaves and branches stood out for their ability to disrupt preexisting TTR fibrils. After narrowing down the chemistry, they identified PGG as the key ingredient driving the effect.
Selective Disruption of TTR Amyloid
In laboratory studies, PGG dismantled amyloid fibrils produced from both mutant and wild-type forms of TTR. Under the same experimental conditions, the compound did not interfere with amyloid-β fibrils linked to Alzheimer’s disease. This selectivity indicates that PGG interacts with TTR aggregates in a specific way rather than broadly attacking all amyloid structures.
The researchers next evaluated PGG in a nematode model (C. elegans) engineered to produce fragments of human TTR. Animals treated with the compound showed fewer protein deposits and experienced significant increases in both lifespan and healthspan. The team also tested amyloid fibrils extracted from the heart tissue of a patient with hereditary TTR amyloidosis. PGG was able to break apart these patient-derived fibrils as well, underscoring its potential relevance in real-world disease.
Structural Insights and Therapeutic Potential
Structural analyses further revealed that multiple galloyl groups attached to a glucose core are essential for activity, providing insight into how the compound interacts with amyloid fibrils at the molecular level.
While further studies are needed to evaluate safety and efficacy in humans, the findings suggest that plant-derived molecules such as PGG could serve as promising lead compounds for next-generation therapies that actively remove pathogenic amyloid deposits, addressing a major unmet need in TTR amyloidosis treatment.
Reference: “Glycosidic scaffold bearing multiple galloyl moieties from pomegranate disrupts transthyretin amyloids” by Asuka Kagami, Nami Hashimoto, Ryoko Sasaki, Yutaro Fukushima, Hari Prasad Devkota, Shoya Tanaka, Mikiyo Wada, Kunitoshi Yamanaka, Shiori Yamakawa, Shogo Misumi, Takeshi Yokoyama, Mineyuki Mizuguchi, Takashi Sato, Teruya Nakamura, Shunsuke Kotani, Mary Ann Suico, Hirofumi Kai, Mitsuharu Ueda and Tsuyoshi Shuto, 20 November 2025, iScience.
DOI: 10.1016/j.isci.2025.114170
Funding: Japan Science and Technology Agency, Kumamoto University
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.