
Life Could Have Emerged From Lakes With High Phosphorus
Life as we know it requires phosphorus. It’s one of the six main chemical elements of life, it forms the backbone of DNA and RNA molecules, acts as the main currency for energy in all cells, and anchors the lipids that separate cells from their surrounding environment.
But how did a lifeless environment on the early Earth supply this key ingredient?
“For 50 years, what’s called ‘the phosphate problem,’ has plagued studies on the origin of life,” said first author Jonathan Toner, a University of Washington research assistant professor of Earth and space sciences.
The problem is that chemical reactions that make the building blocks of living things need a lot of phosphorus, but phosphorus is scarce. A new UW study, published on December 30, 2019, in the Proceedings of the National Academy of Sciences, finds an answer to this problem in certain types of lakes.

The study focuses on carbonate-rich lakes, which form in dry environments within depressions that funnel water draining from the surrounding landscape. Because of high evaporation rates, the lake waters concentrate into salty and alkaline, or high-pH, solutions. Such lakes, also known as alkaline or soda lakes, are found on all seven continents.
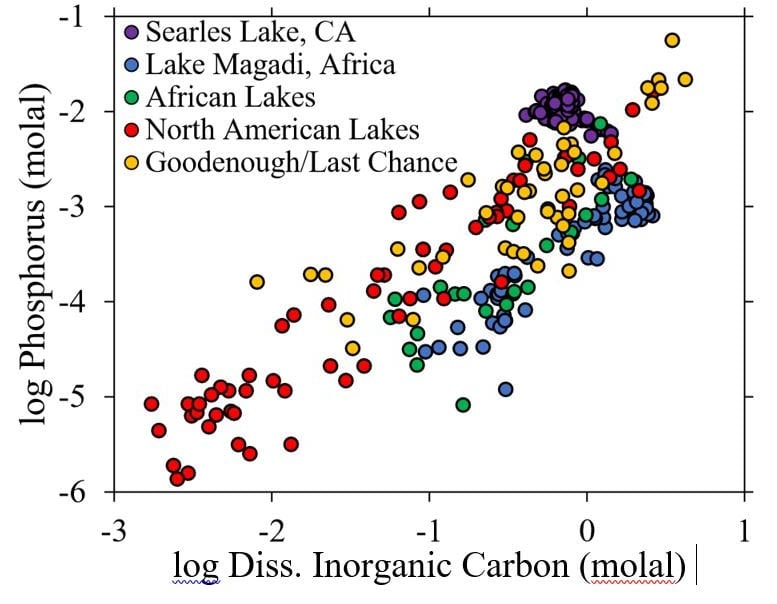
The researchers first looked at phosphorus measurements in existing carbonate-rich lakes, including Mono Lake in California, Lake Magadi in Kenya, and Lonar Lake in India.
While the exact concentration depends on where the samples were taken and during what season, the researchers found that carbonate-rich lakes have up to 50,000 times the phosphorus levels found in seawater, rivers, and other types of lakes. Such high concentrations point to the existence of some common, natural mechanism that accumulates phosphorus in these lakes.
Today these carbonate-rich lakes are biologically rich and support life ranging from microbes to Lake Magadi’s famous flocks of flamingoes. These living things affect the lake’s chemistry. So researchers did lab experiments with bottles of carbonate-rich water at different chemical compositions to understand how the lakes accumulate phosphorus, and how high phosphorus concentrations could get in a lifeless environment.
The reason these waters have high phosphorus is their carbonate content. In most lakes, calcium, which is much more abundant on Earth, binds to phosphorus to make solid calcium phosphate minerals, which life can’t access. But in carbonate-rich waters, the carbonate outcompetes phosphate to bind with calcium, leaving some of the phosphate unattached. Lab tests that combined ingredients at different concentrations show that calcium binds to carbonate and leaves the phosphate freely available in the water.
“It’s a straightforward idea, which is its appeal,” Toner said. “It solves the phosphate problem in an elegant and plausible way.”
Phosphate levels could climb even higher, to a million times levels in seawater, when lake waters evaporate during dry seasons, along shorelines, or in pools separated from the main body of the lake.
“The extremely high phosphate levels in these lakes and ponds would have driven reactions that put phosphorus into the molecular building blocks of RNA, proteins, and fats, all of which were needed to get life going,” said co-author David Catling, a UW professor of Earth & space sciences.
The carbon dioxide-rich air on the early Earth, some four billion years ago, would have been ideal for creating such lakes and allowing them to reach maximum levels of phosphorus. Carbonate-rich lakes tend to form in atmospheres with high carbon dioxide. Plus, carbon dioxide dissolves in water to create acid conditions that efficiently release phosphorus from rocks.
“The early Earth was a volcanically active place, so you would have had lots of fresh volcanic rock reacting with carbon dioxide and supplying carbonate and phosphorus to lakes,” Toner said. “The early Earth could have hosted many carbonate-rich lakes, which would have had high enough phosphorus concentrations to get life started.”
Another recent study by the two authors showed that these types of lakes can also provide abundant cyanide to support the formation of amino acids and nucleotides, the building blocks of proteins, DNA and RNA. Before then researchers had struggled to find a natural environment with enough cyanide to support an origin of life. Cyanide is poisonous to humans, but not to primitive microbes, and is critical for the kind of chemistry that readily makes the building blocks of life.
References:
“A carbonate-rich lake solution to the phosphate problem of the origin of life” by Jonathan D. Toner and David C. Catling, 30 December 2019, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.1916109117
“Alkaline lake settings for concentrated prebiotic cyanide and the origin of life” by J. D.Toner and D. C. Catling, 25 June 2019, Geochimica et Cosmochimica Acta.
DOI: 10.1016/j.gca.2019.06.031
The research was funded by the Simons Foundation’s Collaboration on the Origins of Life.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
13 Comments
Abiogenesis becomes more difficult to believe as we learn more about the processes necessary for life to have evolved.
This article relies on “faith”, speculations, assumptions, and ignorance to rival any religious dogma.
The author needs to turn his collar around and face the overwhelming evidence of “intelligent design”
Statistically there is no other conclusion.
For more information I recommend Jim Trout’s YouTube dissertations.
Biologists don’t “believe” in evolution of life, we see it happen. All your characterization of the article, which simply is a press release of peer published science, is erroneous and without source references. Especially egregious is your claim that someone’s social media publications would constitute – presumably academic – “dissertations”.
On that basis you walk a failed – biology rejects it [ https://en.wikipedia.org/wiki/Intelligent_design ] – religious idea (“intelligent design”) around on a sci/tech site.
Oh, and I forgot this horrendous error: “Statistically there is no other conclusion.”
Full disclosure: I am a bioinformatician, with a first year of *nothing but statistics* (and biology, and software). As an expert I can tell you straight up, *there is only evolution that generates and pass the statistical tests* based on our observations [ https://en.wikipedia.org/wiki/Population_genetics ].
Your “statistically” has never seen a statistic! (Which is presumably why you give no references.)
You cannot prepare the specific dish with ingredients just randomly placed on the table without having proper recipe for it and most importantly, with no cook or precise mechanism capable doing it. Even with all these at hand you still would need proper kitchen accessories to do the job. You cannot create live cat from dead cat and even more: with parts of dead cat tissues. This is not science. More specifically, experiments belong to science but conclusions to mithology or fantasy.
To repeat myself: All your characterization of the article, which simply is a press release of peer published science, is erroneous and without source references.
Instead of the accepted work based on observation, for reasons of your own you offer no alternative whatsoever. That is however not a likely sell.
Finally, modern cosmology tells us *all* structure formation – the entire universe – is a dish that was prepared from random fluctuations in the inflationary field during the era that preceded big bang [ https://www.forbes.com/sites/startswithabang/2019/10/22/what-came-first-inflation-or-the-big-bang/ ]. So your initial claim is generally wrong, and in the case of evolution we have known even earlier – for two centuries now – that there is a simple process, neither fully deterministic (selection) nor fully random (variation and contingency), that produces populations of organisms: evolution. In contrast to the article, and as I lay out in a longer comment currently on hold for acceptance, we know have credible phylogenetic trees extending all the way to the geological root of life.
Oy: “we know” – we now.
I should perhaps also for fuller disclosure list the main mechanisms that fill end empty the population genome variation:
Adding is mutation, genetic flows (such as migration) and recombination (sex).
Paring down is selection, genetic drift (such as population bottlenecks), and inbreeding.
But in general there is a tension between selection and variation in near neutral drift, which is the main mode of genetic loci change.
This is the hard work of bioinformatics, to elucidate the mechanisms. The overall process is on the other hand embarrassingly simple to test, now that sites like NCBI offer freely available genetic data bases based on cheap sequencing, free software and free tutorials. It may take anyone a couple of hours the first time around, but after that one can sit and test – based on observation – evolution in a few minutes. Chose a protein and some species, blast align the sequences, and tree them to see and show descent by modification – simple as that.
NCBI site (in case it is links that makes comment go on hold for approval):
https://www.ncbi.nlm.nih.gov/ .
Good luck with the studies and Happy New Year!
Nice. But the more plausible explanation is an upgrade of sulpher levels after the impact of a comet in a lake
We know from observation that life evolved from a LUCA on the former heat sterile Earth, quick and easy without problem. But where? Biology/geology has over the years narrowed down the locale for the universal ancestor lineage to alkaline hydrothermal vents.
However biochemists may disagree and place the locale as early pools or as here lakes. Never mind that we know there was a global ocean but we don’t know there was any arc islands (say). This paper supports the latter view, but even if the find is interesting the implications are weak.
One reason why the paper is weak is that it cherry picks old papers claiming Hadean Earth had little ocean phosphorylation agents. Later papers have found that this is not so. “Here, we report the occurrence of phosphite in early Archean marine carbonates at levels indicating that this was an abundant dissolved species in the ocean before 3.5 Ga.” [ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3690879/ ]
Phosphite phosporylation does not directly form phosphates, but there is another pathway:
“We know little of the speciation of P in the lithosphere, especially in melts and non-crystalline materials …
Another condensed phosphate, the ring-formed trimetaphosphate ((P3O9)3-), has, on the other hand, been shown to be the most effective condensing agent among polyphosphates, particularly because it can serve as a phosphorylating agent reagent in strongly alkaline aqueous solutions (pH 12) [18,25-28]. Such high pH values can be found in, for instance, the fairly recently discovered modern sediment-starved subduction zones with serpentinite mud volcanoes, like the Mariana Trench [29,30]. …
… Serpentine mud volcanoes similar to those of the Mariana forearc next to the trench occur at Isua, Greenland, and have been dated to early Archean (3.81-3.70 Ga) [31].” [ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4057523/ ]
So we don’t really need acidic lakes and dry/wet cycles to drive phosporylation on early Earth. it looks like it was phosphite (-PO3H) in the oceans .
If we know there was as least much free P in the Hadean oceans as today (since today the biosphere consumes it and locks it away in sediments), the question is only one of degree. Phosphite would make P available to cells, a certain amount of molecules would be phosphate that would make it a slide in with RNA – and this obviously happened – and the question is where we see most phosphates. The free (poly)phosphates would come from the alkaline vents themselves (if any) – different pH – and if so is another support for the main stream theory.
When biochemists say freshwater lakes we ask “where were they”?
When geologists say alkaline hydrothermal vents – after ocean ridges filtering the water with its iron and acids and replaced it with alkali metal rock alkaline conditions (which in turn appears as the iron and hydrogen leaks out fastest in a ocean ridge “conveyor belt”) – we ask “did it happen”?
It most likely did, and we most likely can find out. Freshwater theories, not so much.
Finally, we should also consider that modern phylogenetics makes away with the need to find exact pathways that obsess some areas of astrobiology. Instead we want the most likely set of pathways – a phylogenetic tree.
What a joke… This is not science.
Life comes from life. Not chance molecular combinations over time.
What a joke… This is not about the science.
Evolution is the process of all life, and as noted to some length already it is neither fully deterministic nor fully random, nor are trait trees limited to modern cells. Not personal opinion based on not knowing anything about the subject and great willingness to show such illiteracy.
Life comes from Life? That is the most silly comment I ever had the honor of reading.
Even if you like to deal in make-believe fantasies like Creationism you need to have a start: God made Adam from clay, right!? Non-life to life….
And before that, he/she just created the world and everything from….. Let’s hear it: Nothing!
Or, is God ‘life’ in the sense you try to peddle it: Animal type of life? Than we have an even bigger problem to answer. Where did that ‘life’ come from? Another, bigger God? Hinduism, here we come….
Read the article carefully and note the number of equivocal words.