A new study challenges the long-standing view that Alzheimer’s is driven primarily by amyloid plaques, instead pointing to a subtle but critical competition inside neurons.
New research led by the University of California, Riverside, suggests Alzheimer’s disease may not be driven solely by plaque buildup in the brain, as widely believed. Instead, it may result from one protein disrupting the normal function of another.
For years, scientists have focused on amyloid beta (a-beta) as the main cause of Alzheimer’s. Clusters of this protein are commonly found in patients, and genetic mutations that raise a-beta levels are known to trigger early-onset Alzheimer’s.
However, thousands of clinical trials designed to remove a-beta have failed to stop or reverse the disease.
Researchers also know that another protein, tau, builds up in the brains of people with Alzheimer’s. Still, how tau and a-beta interact has remained uncertain. “In addition to having dementia, Alzheimer’s diagnosis requires both a-beta and tau buildup in the brain,” said UCR chemistry professor and study lead author Ryan Julian. “But many labs focus on the role of one and ignore the other.”
The new study, published in PNAS Nexus points to a direct link between the two proteins.
Tau plays a key role in stabilizing microtubules, which are structures inside cells. These tiny tubes act like transport routes, helping move essential molecules throughout nerve cells. Without them, neurons cannot deliver the materials needed for survival and communication.
A Competition Inside Neurons
The team found that the parts of tau that bind to microtubules closely match the size and shape of a-beta. This similarity suggested that a-beta might also attach to these structures.
To test this, researchers labeled a-beta with a fluorescent marker. Changes in its motion and light signal allowed them to detect when it bound to microtubules.
The results showed that a-beta and tau attach to microtubules with similar strength. This means that when a-beta builds up inside neurons, it can push tau out of place.
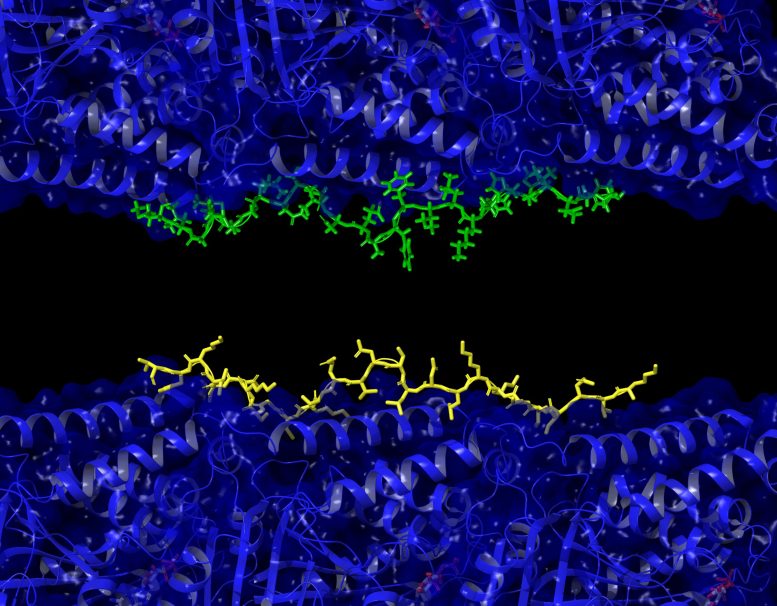
“Our work shows amyloid beta and tau compete for the same binding sites on microtubules, and that a-beta can prevent tau from functioning correctly,” Julian said.
This interference could mark the start of the disease. When tau is displaced, the cell’s internal transport system begins to fail. At the same time, tau can start to clump together and move into parts of the neuron where it does not belong.
The findings suggest that the buildup of a-beta and tau may be a consequence of the disease rather than its original cause. This idea could help explain gaps in earlier theories. For example, plaques that form outside cells may not directly affect tau or the microtubules inside cells.
Aging, Protein Clearance, and New Clues
The model also aligns with evidence that the brain’s protein recycling system slows with age. Autophagy normally removes proteins like a-beta from cells. When this process declines, a-beta can accumulate and compete with tau for access to microtubules.
Other research supports this view. Some studies indicate that lithium may reduce the risk of Alzheimer’s, and earlier work has shown that lithium helps stabilize microtubules. This suggests that strengthening these structures could limit the harmful effects of a-beta.
If confirmed, these findings could change how Alzheimer’s is treated. Instead of focusing only on removing protein clumps, future therapies might aim to block a-beta from interfering with microtubules or improve the cell’s ability to clear it.
Julian said the study brings together many previously disconnected findings.
“This idea helps make sense of many results that previously seemed unrelated,” Julian said. “It gives us a clearer picture of what may be going wrong inside neurons and where new treatments might start.”
Reference: “The microtubule nexus linking amyloid beta and tau: A simple and unifying theory for the underlying cause of Alzheimer’s disease” by Thomas A Shoff, Maxence Derbez-Morin, Peishan Cai and Ryan R Julian, 17 March 2026, PNAS Nexus.
DOI: 10.1093/pnasnexus/pgag034
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Ben 10 reboot