A novel CRISPR-based method links protein isoforms from exon skipping to distinct liver cancer subtypes, offering a potential breakthrough in therapeutic research.
Mutations in our genetic makeup can result in severe problems, such as colon or liver cancer. However, cancer is an intricate disease and the same genetic mutations can result in varying subtypes of tumors in different individuals. Currently, there is no effective method for creating these tumor subtypes in the lab.
Now, Assistant Professor Semir Beyaz from Cold Spring Harbor Laboratory has developed a novel technique for modeling certain subtypes of liver cancer tumors using the CRISPR-Cas9 gene-editing tool.
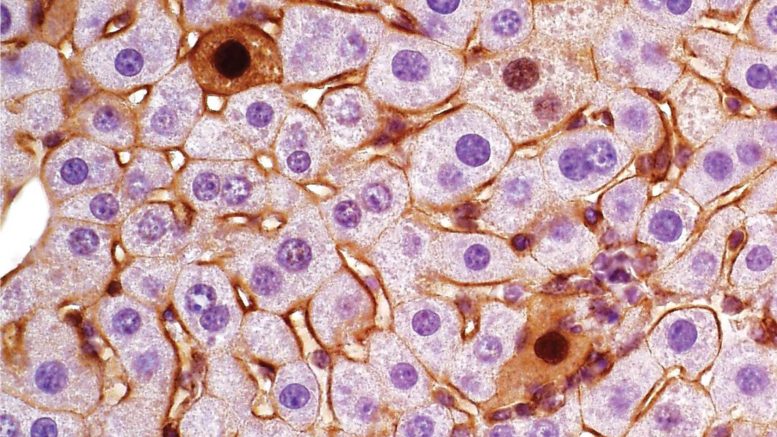
Genes contain the information our bodies need to create proteins. Highly similar proteins produced from the same gene are called isoforms. Different isoforms generate different tumors. This process is known as exon skipping, where multiple parts of a gene are stitched together to make a different version of a protein.
“Everyone thinks that cancer is just one type,” Beyaz explains. “But with different isoforms, you can end up with cancer subtypes that have different characteristics.”
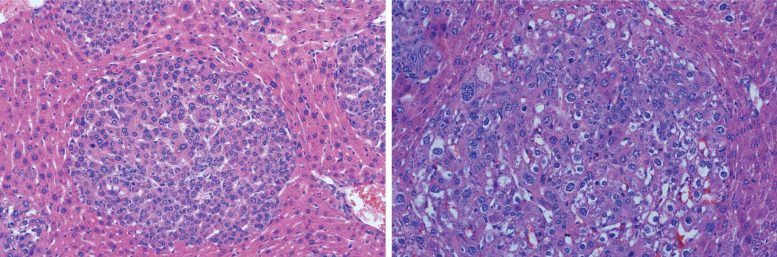
Beyaz and his colleagues produced two distinct tumor subtypes by targeting a single section of the mouse gene, Ctnnb1, with CRISPR. The tool is mostly used to inhibit gene function. This is the first time CRISPR has been used to generate different cancer-causing gain-of-function mutations in mice. These mutations enhance protein activity to promote tumor growth. The team sequenced each tumor subtype to figure out which isoform was associated with the differences they observed.
“We were able to define those isoforms that associated with different cancer subtypes,” Beyaz says. “That was, for us, a surprising discovery.”
Next, to confirm that these isoforms actually caused the variances, they produced them in the mouse without using CRISPR. They found that they were indeed able to generate the two different tumor subtypes with their respective characteristics. Both of these liver tumor subtypes are also found in humans.
The mutations Beyaz targeted can lead to colon and liver cancers. Targeting exon skipping has emerged as a potential therapeutic approach for treating cancer and other diseases. Beyaz’s new study method allows researchers to investigate this phenomenon in living mice cells using CRISPR. The platform could someday help researchers develop new therapeutic interventions. “Ultimately,” Beyaz explains, “what we want to do is find the best models to study the biology of cancer so that we can find a cure.”
Reference: “CRISPR-induced exon skipping of β-catenin reveals tumorigenic mutants driving distinct subtypes of liver cancer” by Haiwei Mou, Onur Eskiocak, Kadir A. Özler, Megan Gorman, Junjiayu Yue, Ying Jin, Zhikai Wang, Ya Gao, Tobias Janowitz, Hannah V. Meyer, Tianxiong Yu, John E Wilkinson, Alper Kucukural, Deniz M. Ozata and Semir Beyaz, 15 January 2023, The Journal of Pathology.
DOI: 10.1002/path.6054
Funding for the study was from the National Cancer Institute, the National Institutes of Health, the Mark Foundation For Cancer Research, the Oliver S. and Jennie R. Donaldson Charitable Trust, the STARR Cancer Consortium, the National Center for Advancing Translational Sciences (NCATS), and the Swedish Research Council.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.