
The glass structure of a material is often believed to mimic its corresponding liquid. Polyamorphism between ices has been used as a guide to elucidate the properties of liquid water. But how many forms of amorphous ices are there? Do we understand how metastable high-pressure crystalline ice evolves toward the thermally stable low-density form? An international research team led by Chuanlong Lin and Wenge Yang from HPSTAR and John S. Tse from the University of Saskatchewan has revealed a multiple-step transformation mechanism using state-of-the-art time-resolved in situ synchrotron x-ray diffraction. A temperature/time-dependent kinetic pathway with three distinctive transitions was identified in the structural evolution from metastable crystalline ice (ice VII or ice VIII) to the thermodynamically stable ice I. These intermediate processes compete against each other. The end result is a juxtaposition of these processes. The work is published in the scientific journal PNAS.
Water plays a vital role in the origin of life on Earth. In the liquid phase, it exhibits many unusual properties. In the solid phase, ordinary ice also displays diverse phase transitions at high pressure. Many theoretical and experimental studies have been devoted to understanding the underlying inter-conversion mechanisms. So far, most experiments have been ex situ measurements on recovered samples and lack detailed information on the structural evolution accompanying the transformation. Previous studies have been hindered by technical difficulties in monitoring the rapid structural change over a broad pressure and temperature range.
In 2017, Lin and his colleagues overcame the experimental challenge. A series of studies was conducted to investigate ice transitions by combining in situ time-resolved x-ray diffraction, and remote pressure control with different ramp rates within a low-temperature cryostat. This capability allowed the suppression of thermally-driven crystalline-crystalline transitions [PNAS 115, 2010-2015(2018)]. Important insights into the complexity of the poly-amorphous transformations were obtained, such as the kinetically-controlled two-step amorphization in ice Ih [Phys. Rev. Lett. 119, 135701(2017)] and the successful venture into the no man’s land [Phys. Rev. Lett. 121, 225703(2018)].
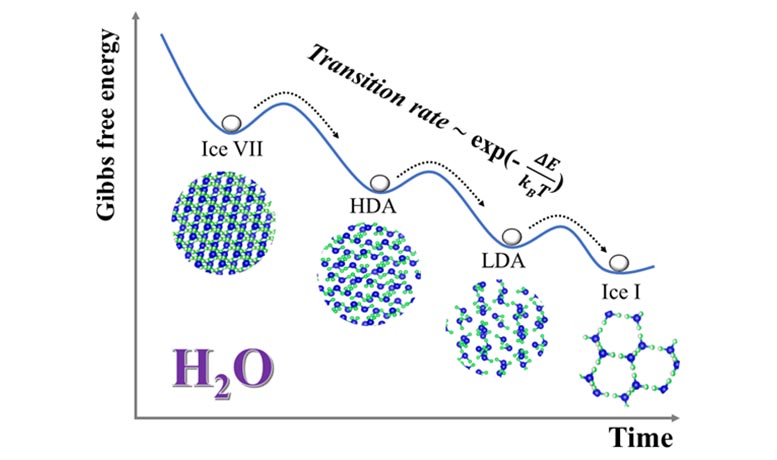
Now, they try to answer what exactly is the nature of the amorphous-amorphous phase transformation processes? Using the newly developed techniques, they explored the “mirror” process, i.e., reverse transformation from a meta-stable high-density crystalline ice (i.e, ice VII or ice VIII) to the ambient stable ice I. They identified the temperature/time-dependent kinetic pathways and characterized the interplay/competition between the high density amorphous (HDA)-low density amorphous (LDA) transition and recrystallization. Contrary to previously reported ice VII (or ice VIII) — LDA — ice I transformation sequences, time-resolved measurements show a three-step process: initial transformation of ice VII to HDA, followed by a HDA — LDA transition, and then crystallization of LDA into ice I. Both the amorphization of ice VII and the HDA to LDA transition show distinctive thermal activation mechanisms. Significantly, both processes exhibit the Arrhenius behavior with a temperature-dependent duration time (τ) and a ‘transition’ temperature at around 110-115 K.
Large-scale molecular-dynamics calculations also support their experimental findings. Furthermore, it shows the HDA to LDA transformation is continuous with a large density difference and involves substantial displacements of water in the nano-scale. This study presents a new perspective on the metastability and complexities in shaping ice-transition kinetic pathways.
Reference: “Temperature-dependent kinetic pathways featuring distinctive thermal-activation mechanisms in structural evolution of ice VII” by Chuanlong Lin, Xuqiang Liu, Xue Yong, John S. Tse, Jesse S. Smith, Niall J. English, Bihan Wang, Mei Li, Wenge Yang and Ho-Kwang Mao, 22 June 2020, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2007959117
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.