
Scientists can now see a hidden battery ingredient — and it could supercharge how fast and how long lithium-ion batteries last.
Scientists at the University of Oxford have created an advanced technique that allows them to clearly see a crucial but previously hard-to-detect part of lithium-ion battery electrodes. The findings, published today (February 17) in Nature Communications, could improve how battery electrodes are manufactured and lead to faster charging and longer-lasting Li-ion batteries.
The research centers on polymer binders used in the negative electrodes of lithium-ion batteries (anodes). Although these binders account for less than 5% of the electrode’s weight, they play an essential role in keeping the electrode intact. They influence mechanical strength, electrical and ionic conductivity, and overall battery lifespan. Because binders lack distinctive visual features and are present in such small amounts, scientists have struggled to track where they are located inside the electrode. That limitation has made it difficult to fine-tune battery performance, since binder placement directly affects conductivity, structural stability, and durability over time.

Patent Pending Staining Technique Makes Binders Visible
To solve this long-standing challenge, the team developed a patent-pending staining method that attaches traceable silver and bromine markers to widely used cellulose- and latex-based binders in both graphite- and silicon-based anodes. Once tagged, the binders can be detected because they emit characteristic X-rays (measured with energy-dispersive X-ray spectroscopy) or reflect high-energy electrons from the surface (measured with energy-selective backscattered electron imaging). Under an electron microscope, these signals reveal detailed information about where elements are distributed and what the surface structure looks like.
Lead author Dr. Stanislaw Zankowski (Department of Materials, University of Oxford) said: “This staining technique opens up an entirely new toolbox for understanding how modern binders behave during electrode manufacturing. For the first time, we can accurately see the distribution of these binders not only generally (i.e., their thickness throughout the electrode), but also locally, as nanoscale binder layers and clusters, and correlate them with anode performance.”
The approach works not only for traditional graphite electrodes but also for more advanced materials such as silicon or SiOx. This makes the method useful for current lithium-ion batteries as well as next-generation designs.
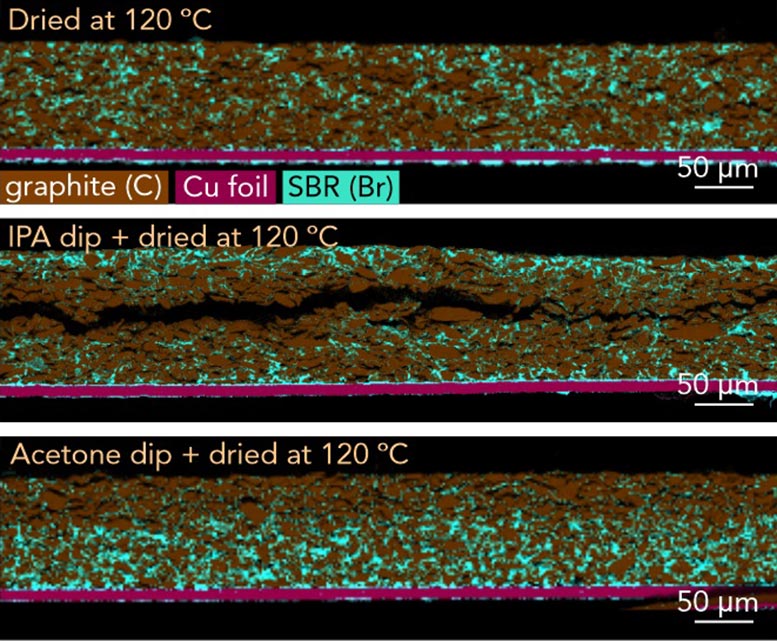
Small Manufacturing Changes Cut Resistance by 40%
Using this imaging technique, the researchers discovered that even slight differences in binder distribution can significantly influence charging speed and battery lifespan. By refining slurry mixing and drying steps during electrode production, they were able to lower the internal ionic resistance of test electrodes by as much as 40% – a major barrier to faster charging.
The team also captured detailed images of extremely thin layers of carboxymethyl cellulose (CMC) binder coating graphite particles. The method allowed them to detect CMC films just 10 nm thick and observe structures spanning four orders of magnitude within a single image. These images showed that what begins as a uniform CMC coating can break apart into uneven, patchy fragments during processing, which may weaken battery performance and long-term stability.
Co-author Professor Patrick Grant (Department of Materials, University of Oxford) said: “This multidisciplinary effort-spanning chemistry, electron microscopy, electrochemical testing, and modelling- has resulted in an innovative imaging approach that will help us to understand key surface processes that affect battery longevity and performance. This will drive forward advancements across a wide range of battery applications.”

Reference: “Chemical staining for fundamental studies and optimization of binders in Li-ion battery negative electrodes” by Stanislaw P. Zankowski, Samuel Wheeler, Thomas Barthelay, Wai Man Chan, Michael Metzler and Patrick S. Grant, 17 February 2026, Nature Communications.
DOI: 10.1038/s41467-026-69002-1
The work was supported by the Faraday Institution’s Nextrode project and has already drawn strong interest from industry, including major battery manufacturers and electric vehicle companies.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.