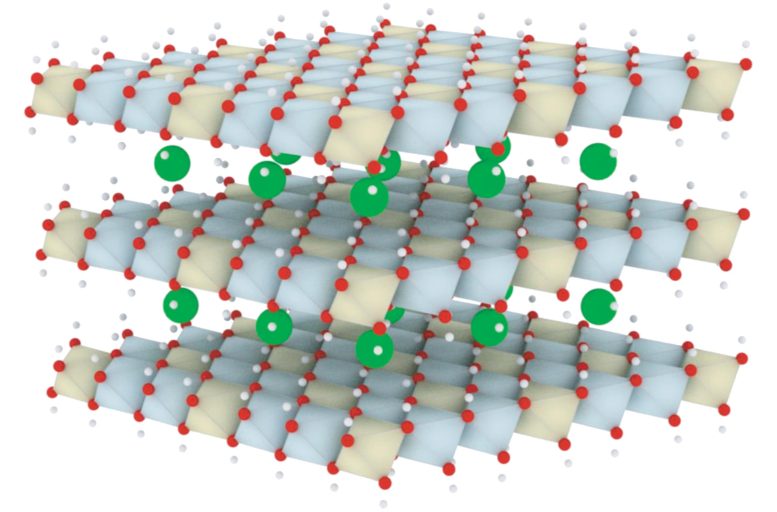
New achievements of layered double hydroxides toward efficient, stable, fast oxygen evolution reaction.
Scientists in China have made advancements in the use of layered double hydroxides to improve oxygen evolution reaction (OER) energy utilization, toward the goal of efficient hydrogen generation for renewable energy. The researchers, from the Beijing University of Chemical Technology, recently published their work in the journal Energy Material Advances.
“With the rising demand and consumption of fossil fuels, energy shortage and environmental pollution are becoming severe and unignorable,” said the corresponding author Mingfei Shao, professor with the State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing. “It is necessary to explore sustainable and renewable energy, especially, hydrogen is a new energy with splendid application prospects.”
Production of highly pure hydrogen can be achieved by electrochemical water splitting using the electricity transformed from renewable energy such as wind and solar energy. But as one of the half-reactions, OER is a four-electron process, which causes a low efficiency of energy utilization, according to Shao.
Layered Double Hydroxides in Oxygen Evolution Reaction
Shao and his team focus on layered double hydroxides (LDHs), an important kind of two-dimensional materials with wide tunability of the types, molar ratios of the metal elements, and the nature of interlayer anions. They are outstanding catalysts for OER in alkaline medias.
“We summarized four common strategies applied to improve the OER performance of LDHs. Through these strategies, the overpotential of OER can be decreased obviously, leading to a high efficiency of energy utilization,” Shao said. “Some works about the identification of active sites for LDHs are introduced. Revelation of reaction mechanism and active sites provide the theoretical guidance to design efficient electrocatalysts.”
Until now, the development and exploration of OER catalysts is mostly in the experimental stage, which cannot meet the standards for large-scale practical use. For example, there are still problems with enlarging the size of catalysts and maintaining stability during OER with a severe condition, such as high alkalinity and high temperature. Besides, most reported preparation methods of LDH-based catalysts are complicated and time-consuming, which brings high costs and restricts their application as well as popularization, according to Shao.
“The recognition of reactive oxygen species such as oxygen species adsorbed by active sites on the surface of electrocatalysts and oxygen radical dispersed in the solution during OER still remains ambiguous due to the instable and unapparent existence of reactive oxygen species,” Shao said. “After recognizing these reactive oxygen species, how to take advantage of them for more efficient OER is still vital.”
“We hope this review can offer ideas to further identify the active sites for LDHs with the purpose of providing guidance to design more advanced electrocatalysts towards electrochemical water splitting,” Shao said.
Reference: “Layered Double Hydroxides for Oxygen Evolution Reaction towards Efficient Hydrogen Generation” by Xin Wan, Yingjie Song, Hua Zhou and Mingfei Shao, 7 September 2022, Energy Material Advances.
DOI: 10.34133/2022/9842610
Other contributors include Xin Wan, Yingjie Song, and Hua Zhou, all with the State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing.
The National Natural Science Foundation of China (22090031, 21922501 and 21871021), Project funded by China Postdoctoral Science Foundation (2021M690319) supported this work.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Any sort of isocyanates will give a positive reaction but what about the toxic vapours from them