
Water plays a crucial role in boosting the conversion of polyolefins into valuable fuels when paired with ruthenium catalysts, presenting a promising approach to addressing global plastic waste.
Plastics are incredibly versatile materials that have become integral to nearly every aspect of modern life. However, with global plastic production now exceeding 400 million tons annually, the environmental impact of plastic waste has reached critical levels. The majority of plastic waste—nearly 90%—is not recycled, amplifying the pollution crisis. To address this growing challenge, innovative technologies are urgently needed.
Catalytic recycling techniques, such as hydrogenolysis and hydrocracking, offer a promising solution. These advanced chemical processes use catalysts to break down plastic waste into simpler, high-value components like chemicals and fuels. Unlike traditional recycling, which involves melting and remolding plastics into lower-quality products, catalytic recycling enables more efficient and sustainable reuse by creating materials with greater economic and environmental value.
While catalytic recycling shows significant potential, further development and refinement are necessary before it can be implemented on a large industrial scale.
A Breakthrough in Polyolefin Recycling
In a recent study published in Nature Communications, a research team led by Professor Insoo Ro of Seoul National University of Science and Technology, Korea, recently made a breakthrough discovery in the catalytic recycling of polyolefins, which comprise 55% of global plastic waste. As explained in their article, the researchers revealed the surprising benefits of adding water during polyolefin depolymerization when using ruthenium (Ru)-based catalysts.
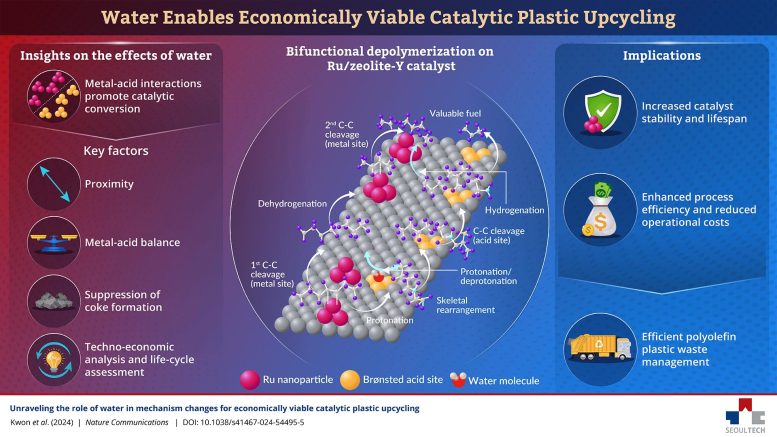
After synthesizing and experimenting on various Ru-based catalysts on different supports, the team found that catalysts with both metal and acid sites exhibit dramatically improved conversion rates when water is added to the reaction mixture. “The addition of water alters the reaction mechanisms, promoting pathways that enhance catalytic activity while suppressing coke formation,” explains Dr. Ro, “This dual role improves process efficiency, extends catalyst lifespan, and reduces operational costs.”
The researchers investigated the reaction mechanisms in detail, shedding light on the effect of Ru content and the proximity and balance between metal and acid sites. Under optimal conditions, Ru/zeolite-Y catalysts showcased a 96.9% conversion rate for polyolefins.
A Viable Alternative to Conventional Waste Management
Finally, to explore the viability of this type of catalytic recycling, the team conducted a techno-economic analysis and a life cycle assessment of the proposed approach. The results clearly underscored the potential of implementing a real commercial-scale process using Ru/zeolite-Y catalyst.
“The addition of water not only enhances carbon efficiency, it improves economic and environmental performance, also increases the conversion of polyolefins to valuable fuels like gasoline and diesel,” highlights Dr. Ro. Adding further, he says, “This approach thus represents a viable alternative to conventional waste management practices and offers a solution to reduce landfill and ocean pollution caused by polyolefins—the largest contributor to plastic waste.”
Overall, this breakthrough in catalytic depolymerization could revolutionize how we deal with plastic pollution and help us efficiently deal with this serious environmental threat. The research team has high hopes that this technology will evolve over the next few years to the point that mixed plastic waste can be processed without pre-sorting, making recycling efforts more cost-effective and simpler to implement.
“By demonstrating a sustainable and economic approach to transforming plastic waste into valuable resources, our research could help drive policy changes, inspire investment in advanced recycling infrastructure, and foster international collaborations to address the global plastic waste crisis. Over time, these advancements promise cleaner environments, reduced pollution, and a more sustainable future,” concludes Dr. Ro on an optimistic note.
Reference: “Unraveling the role of water in mechanism changes for economically viable catalytic plastic upcycling” by Taeeun Kwon, Byeongchan Ahn, Ki Hyuk Kang, Wangyun Won and Insoo Ro, 29 November 2024, Nature Communications.
DOI: 10.1038/s41467-024-54495-5
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
How is ruthenium a “viable” option? It’s a pretty rare and expensive element, found mostly in just two places on Earth. This is just another junk science piece of fluff someone had an interest in pushing.
So, some guys have found something in a lab. Great! They called it a “breakthrough”. Wonderfu!! The solution to our problems is finally at hand. Marvelous! Now it only needs to be translated from the lab into a real world implementation and scaled up… All of a sudden – crickets.
Nothing to see here, move on.
I have always wandered about being able to just all plastic no matter what kind and shred it, add a resin that is compatible with all types, add ultra violet protection, then extrude it into different shapes to make landscape blocks and such. Most important is can a resin be made to stop plastic breaking down?
How about using glass bottles and jars and governments ensuring sellers put a $1 deposit on them to ensure their return? It used to work, then some idiots invented tetrapaks and polycabonate containers. That was in the 1970s, where all bad things came from.